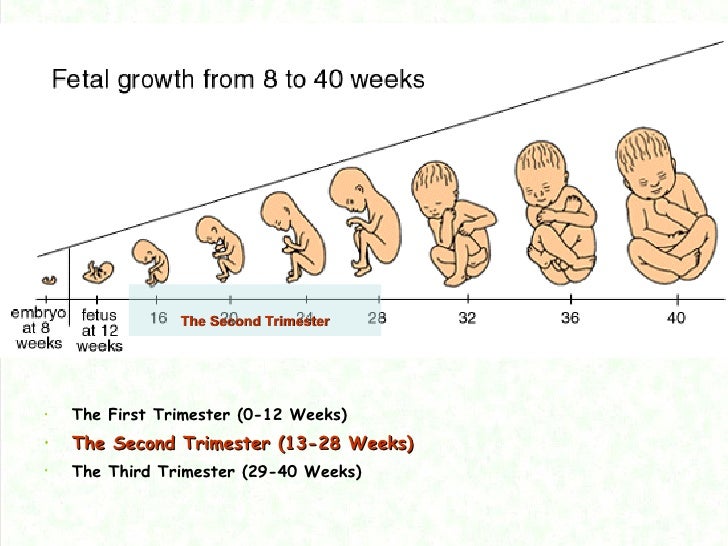
La semana 10 de embarazo se encuentra en la mitad del tercer mes, llegando ya casi al final del crítico primer trimestre.
En este momento, se inicia la etapa fetal, es decir, el embrión pasa a denominarse feto, el cual ya tiene todos sus sistemas básicos formados.
En cuanto a la mujer, su embarazo aún pasa desapercibido para el resto de la gente a las 10 semanas, pero ella ya puede notar el vientre hinchado y un ligero aumento de peso.
A continuación tienes un índice con los 8 puntos que vamos a tratar en este artículo.
Índice
1.
Crecimiento del feto
2.
Síntomas en la madre
3.
Control del embarazo
3.1.
Análisis de sangre
3.2.
Cribado combinado del primer trimestre
4.
Cuidados
5.
Preguntas de los usuarios
5.1.
¿Cómo puedo saber si tengo un embarazo de gemelos o mellizos en la semana 10?
5. 2.
¿Cuántos meses son 10 semanas de embarazo?
5.3.
¿Cómo está el bebé a las 10 semanas de embarazo?
6.
Lectura recomendada
7.
Bibliografía
8.
Autor
En la semana 10 de embarazo, el embrión pasa a llamarse feto hasta la fecha de su nacimiento. Mide aproximadamente 3-4 cm y su peso es de unos 5 gramos.
El cuerpo del feto ya está más alargado y empieza a parecerse más a un humano. Además, es muy activo: mueve sus piernas dando patadas y traga líquido amniótico de forma constante.
Otros cambios importantes en el feto durante esta décima semana son los siguientes:

En la décima semana de embarazo, es posible que los síntomas molestos de las primeras semanas, como las náuseas y vómitos, empiecen a remitir.
Todavía no se han producido importantes cambios en el cuerpo de la mujer, pero empezará a notar que la ropa le está más ajustada por la zona de la cintura. El útero ha aumentado considerablemente de tamaño y ocupa gran parte de la pelvis.
Por otra parte, un cambio importante en esta semana es que los pechos aumentan de tamaño. Es importante comprar un sujetador especial para gestantes llegados a este punto, pues ayudará a evitar problemas de espalda.
Es importante comprar un sujetador especial para gestantes llegados a este punto, pues ayudará a evitar problemas de espalda.
Otros síntomas característicos en la semana 10 de embarazo son los siguientes:
Por último, el estado de ánimo suele mejorar a partir de esta semana gracias a la adaptación del cuerpo al estado de gestación y a la disminución de síntomas molestos.
La mayoría de embarazadas afirman que realmente empiezan a disfrutar del embarazo a partir de la semana diez.
Es importante hacer una primera visita al ginecólogo si aún no lo has hecho en la semana 10. En la ecografía será posible tomar las medidas del feto, visualizar las estructuras fetales y escuchar su latido cardiaco.
A partir de este momento empieza el seguimiento del embarazo. El ginecólogo tiene en cuenta todos los antecedentes de la mujer para incluirlos en su historia: embarazos previos, posibles abortos, síntomas destacados, etc.
También es muy importante controlar el peso y la presión arterial de la madre, medidas que se tomarán en cada revisión ginecológica.
En función de las características del embarazo, edad de la madre, riesgo de aborto u otros aspectos, será necesario llevar un control más o menos estricto de la gestación. No obstante, las pruebas que comentamos a continuación sí que se hacen en todos los embarazos hoy en día.
Durante el transcurso del primer trimestre del embarazo, es necesario hacer un análisis de sangre con el objetivo de comprobar los siguientes parámetros:
En caso de obtener algún resultado alterado, el ginecólogo tomará las medidas oportunas para que no exista ningún riesgo para la madre o el feto. Es muy importante tratar las infecciones para que éstas no pasen al feto.
En este primer análisis de sangre también se determinan otras proteínas necesarias para hacer el llamado screening bioquímico que comentaremos a continuación.
¡Ya somos 18.308!
Únete a la comunidad
Reproducción Asistida ORG
Esta prueba diagnóstica prenatal también se conoce como triple screening y suele hacerse entre las semanas 10 y 12 de embarazo.
Consiste en la estimación de las probabilidades de riesgo de que el feto esté afectado por síndrome de Down u otras cromosomopatías.
Para hacer este cribado combinado, es necesario hacer una extracción de sangre materna y una ecografía para obtener los siguientes parámetros:
Normalmente, primero se hace el análisis de sangre sobre la semana 8 u 11 y, a continuación, la ecografía en la semana 12 de gestación. No obstante, hay casos en que ambos estudios se hacen conjuntamente.
No obstante, hay casos en que ambos estudios se hacen conjuntamente.
Para hacer el cálculo de la probabilidad de riesgo que hemos comentado, se toman en cuenta estos 3 parámetros junto con la edad y el peso de la embarazada en el momento de hacer los análisis y la ecografía.
Puesto que esta prueba solamente es una estimación, si se obtuviera un resultado que indique posibilidad de riesgo, es recomendable hacer una amniocentesis o biopsia de corión para confirmar que efectivamente existe alguna alteración cromosómica.
A partir de este momento de la gestación, la mujer va a empezar a notar más el crecimiento del feto en su interior, ya sea por el aumento de peso o por las molestias en la espalda. Es muy importante practicar deporte apto para embarazadas para mantener el buen estado físico y combatir los posibles dolores.
La alimentación también es clave para el correcto desarrollo del bebé, pues éste toma los nutrientes, vitaminas y minerales de la madre a través de la sangre que fluye por la placenta y el cordón umbilical.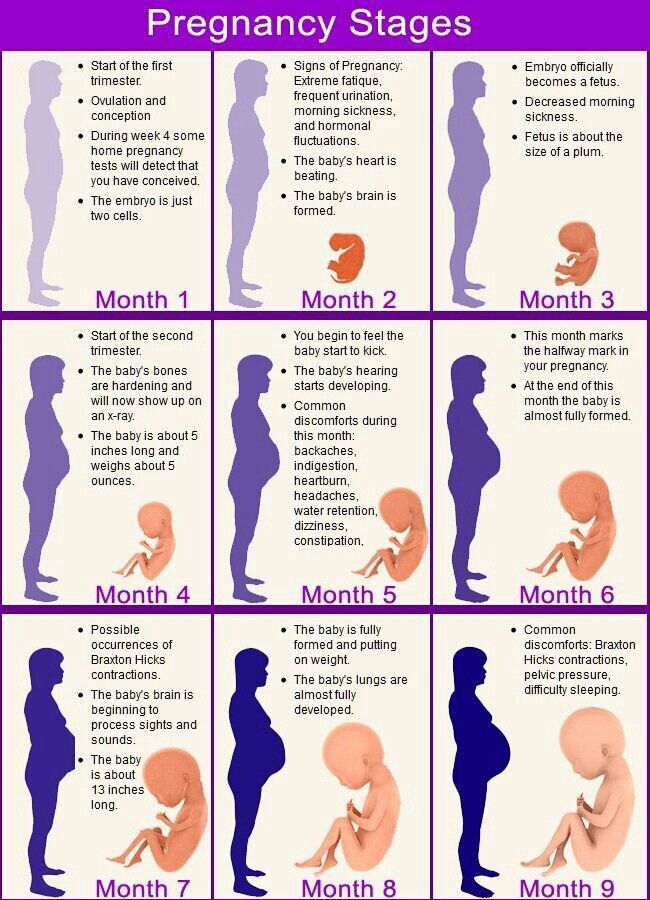
A continuación, vamos a comentar todas la recomendaciones para un embarazo saludable a partir de la semana 10:
Por Zaira Salvador (embrióloga).
Lo más recomendable es hacer una ecografía de ultrasonido para poder ver el número de sacos gestacionales y fetos que hay. También será posible escuchar el latido de dos fetos en la ecografía.
También será posible escuchar el latido de dos fetos en la ecografía.
Por otra parte, las mujeres con embarazo múltiple notan un mayor aumento de peso al llegar a la semana 10, así como la elevación de la parte inferior del vientre. Los síntomas también suelen ser más notorios que en el resto de mujeres: pechos más sensibles, más náuseas, etc.
Por Zaira Salvador (embrióloga).
Las 10 semanas de embarazo se corresponden con el tercer mes. Exactamente, la mujer estaría de 2 meses y medio.
No obstante, cabe señalar que la edad gestacional del feto es de 8 semanas, es decir, de 2 meses.
Por Zaira Salvador (embrióloga).
El cuerpo del bebé ya está formado con diez semanas de gestación y se asemeja al de los humanos. Mide entre 3 y 4 cm, tiene una cabeza grande y los brazos y las piernas ya se han alargado. También es posible intuir sus rasgos faciales: se han formado los ojos aunque están cerrados por los párpados, la nariz es más prominente y las orejas y la boca también se han formado.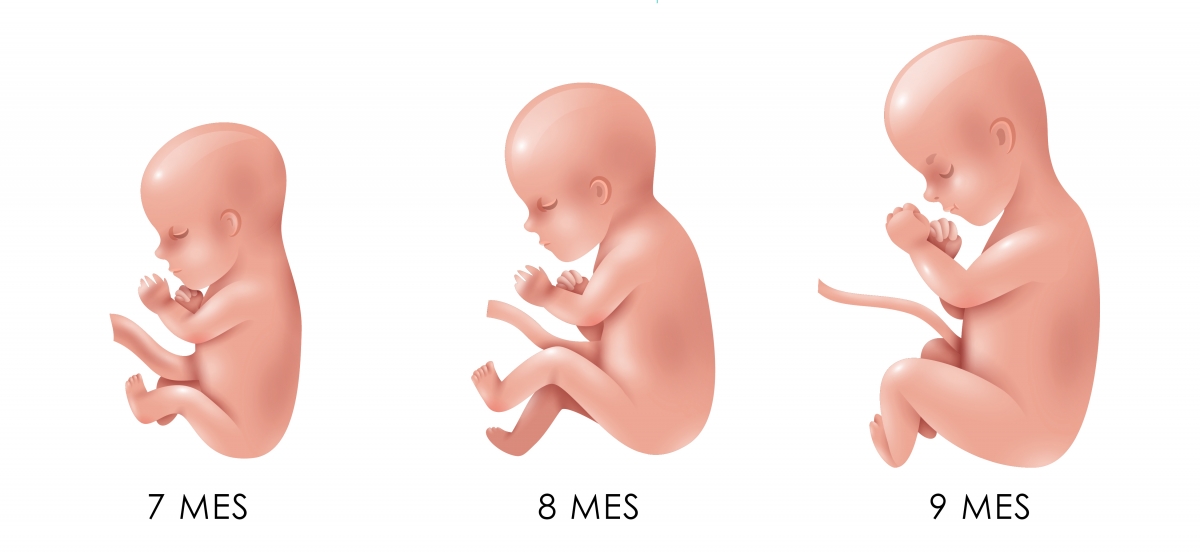 Por último, el bebé ya puede mover sus extremidades y abrir y cerrar las manos.
Por último, el bebé ya puede mover sus extremidades y abrir y cerrar las manos.
Si estás embarazada del 10 semanas, te interesará leer el siguiente artículo próximamente: Once semanas de embarazo.
Un sangrado en la semana 10 de embarazo puede ser indicativo de diferentes cosas. Te recomendamos leer el siguiente post para obtener información más detallada: ¿Es normal sangrar en el embarazo?
Icons / List-viewCreated with Sketch.
Temas en este artículo
Icons / arrow-downCreated with Sketch.
 ..
..Te explicamos cómo evoluciona tu cuerpo a las 10 semanas de embarazo y cómo va creciendo tu bebé. Te ayudamos a despejar todas tus dudas.
El tamaño de tu bebé esta semana
Ciruela pasa
La semana 10 de embarazo se trata de la última semana que tiene lugar en la etapa embrionaria. Por tanto, cuando esta finalice, comenzará una nueva etapa llamada fetal. Es por ello que, a partir de la semana 11 de gestación, el embrión pasará a denominarse feto hasta que se produzca su nacimiento.
Diversos cambios tienen lugar a lo largo de la semana 10 entre los que podemos destacar:
 Además, la frente presenta amplia superficie con respecto al resto de su cara, debido al desarrollo cerebral que está experimentando.
Además, la frente presenta amplia superficie con respecto al resto de su cara, debido al desarrollo cerebral que está experimentando.
Embarazada de 10 semanas
La semana 10 de embarazo corresponde a la edad del embrión de 8 semanas de desarrollo y tiene una CRL aproximada de unos 35-43 milímetros.
Ahora, te vamos a mostrar cómo es el embrión en la semana 10, a través de una ecografía y un fantástico vídeo que te sorprenderá.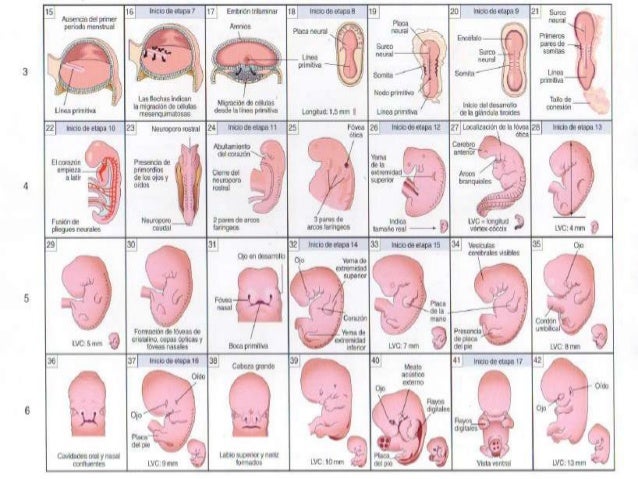 ¡Mira!
¡Mira!
Ecografía de 10 semanas
Llegó la semana 10 y con ella el momento de realizarte tu primer control analítico de sangre y orina. Pero, ¿cuál es el objetivo de este primer análisis? Con la analítica de sangre se busca controlar los siguientes parámetros:

Por otro lado, el objetivo principal de la analítica de orina será el de detectar precozmente una posible infección de orina que no manifieste síntomas en la mujer, algo que es frecuente en el embarazo, por lo que se repetirá en cada trimestre.
Como hemos comentado, durante el embarazo, hay más riesgo de sufrir infecciones de orina, por lo que es importante que sepas detectar algunas señales de alarma que te puedan hacer sospechar como:
En ese caso, no te asustes, es algo muy común durante la gestación. No obstante, es importante que lo consultes lo antes posible con tu matrona o, en caso de coincidir con un control ecográfico, con tu obstetra. En el caso de que se confirme dicha infección, es fácil de tratar con antibióticos compatibles con el embarazo.
Entre los consejos que más pueden ayudarte en la semana 10 de embarazo podemos encontrar:
 Es importante, ya que todo lo que tú ingieras será transportado a tu bebé a través de la placenta y del cordón umbilical.
Es importante, ya que todo lo que tú ingieras será transportado a tu bebé a través de la placenta y del cordón umbilical.

SUPERVISIÓN CIENTÍFICA: Federación Asociaciones de Matronas de España – FAME
Esther Baeza Pereñíguez.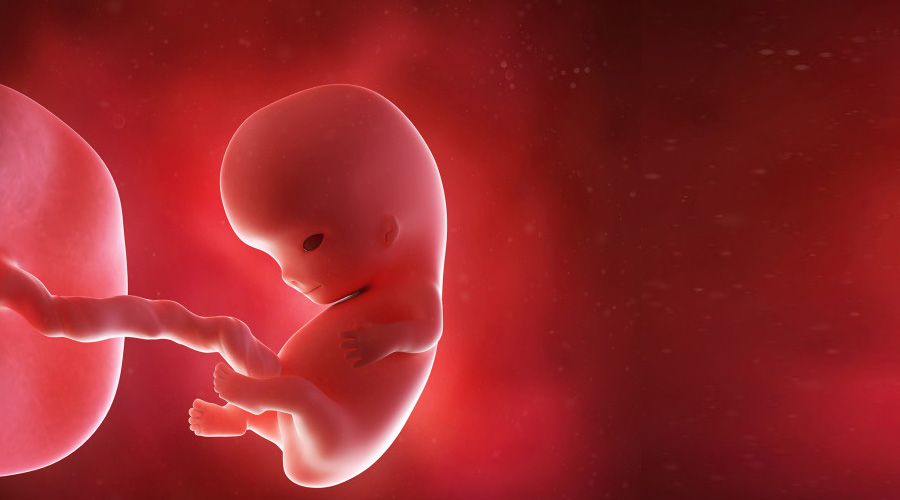 Matrona C.S. Los Alcázares (Murcia).
Matrona C.S. Los Alcázares (Murcia).
Minerva Morcillo Múñoz. Matrona Servicio Murciano de Salud.
……….
Es lo más normal del mundo. Te explicamos por qué te pasa.
¡Descubre por qué sientes ganas de orinar con frecuencia!
Te explicamos qué tienes que hacer si se presentan contracciones en el primer trimestre, y cuándo debes acudir al médico.
¡Descubre qué hacer si crees que tienes contracciones!
¿Eres toda una yoguini o quieres empezar a hacer yoga durante el embarazo? Sea como sea, tu profesor te orientará, pero no está de más que leas esto…
¡Descubre qué posturas de yoga debes evitar!
Son esos deseos irresistibles de comer algún alimento en especial.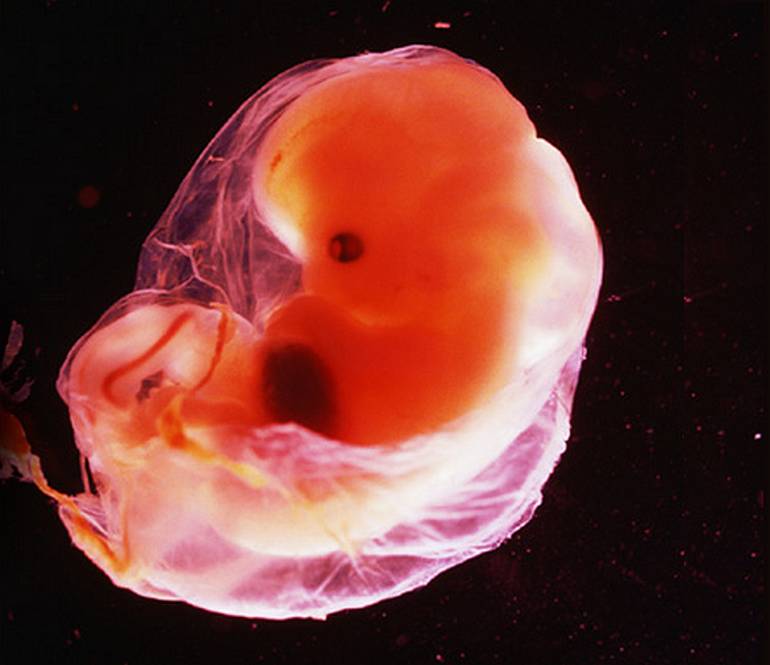 Pero, ¿son un mito o una realidad?
Pero, ¿son un mito o una realidad?
¡Descubre qué son los “antojos” en realidad!
………
Y ahora, haz clic sobre la imagen y mira tu DIBU correspondiente a la semana 10 de embarazo.
¡Te enganchará!
T. L. Botasheva,
A. V. Khloponina,
V. V. Vasilyeva,
O. P. Zavodnov,
L. V. Kaushanskaya,
E. V. Zheleznyakova
https://doi.org/10.21886/2219-8075-2018-9-3-70-76
PDF (Rus)
|
HTML
|
XML
Purpose: identification of seasonal features of melatonin metabolism and hormonal status in pregnant women depending on the sex of the fetus.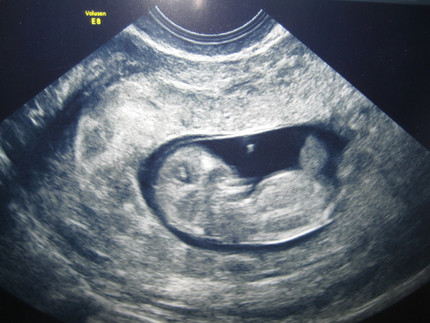 Materials and methods: 538 women were examined in the period of physiological gestation of 37-40 weeks. The first group – 286 pregnant women with female fetuses, the second group – 254 pregnant women with male fetuses. Assessment of hormonal status included the determination of the level of cortisol, estradiol, ACTH, progesterone, testosterone in the blood and melatonin in the urine of pregnant women. nine0021 Results: The level of 6-sulfatoxymelatonin in morning urine, regardless of the sex of the fetus, is higher in seasons with a predominance of dark time of the day. The absolute levels of 6-sulphate-oxy-melatonin in the urine of all women at 37-40 weeks of gestation were the lowest in the light-saturated summer and spring months, while in the winter the values were maximum. Melatonin production is statistically significantly higher in pregnant women with female fetuses in autumn and winter. Regardless of the time of year, absolute levels of estradiol and progesterone were found to be higher in pregnant women with female fetuses than in the case of male fetuses.
Materials and methods: 538 women were examined in the period of physiological gestation of 37-40 weeks. The first group – 286 pregnant women with female fetuses, the second group – 254 pregnant women with male fetuses. Assessment of hormonal status included the determination of the level of cortisol, estradiol, ACTH, progesterone, testosterone in the blood and melatonin in the urine of pregnant women. nine0021 Results: The level of 6-sulfatoxymelatonin in morning urine, regardless of the sex of the fetus, is higher in seasons with a predominance of dark time of the day. The absolute levels of 6-sulphate-oxy-melatonin in the urine of all women at 37-40 weeks of gestation were the lowest in the light-saturated summer and spring months, while in the winter the values were maximum. Melatonin production is statistically significantly higher in pregnant women with female fetuses in autumn and winter. Regardless of the time of year, absolute levels of estradiol and progesterone were found to be higher in pregnant women with female fetuses than in the case of male fetuses.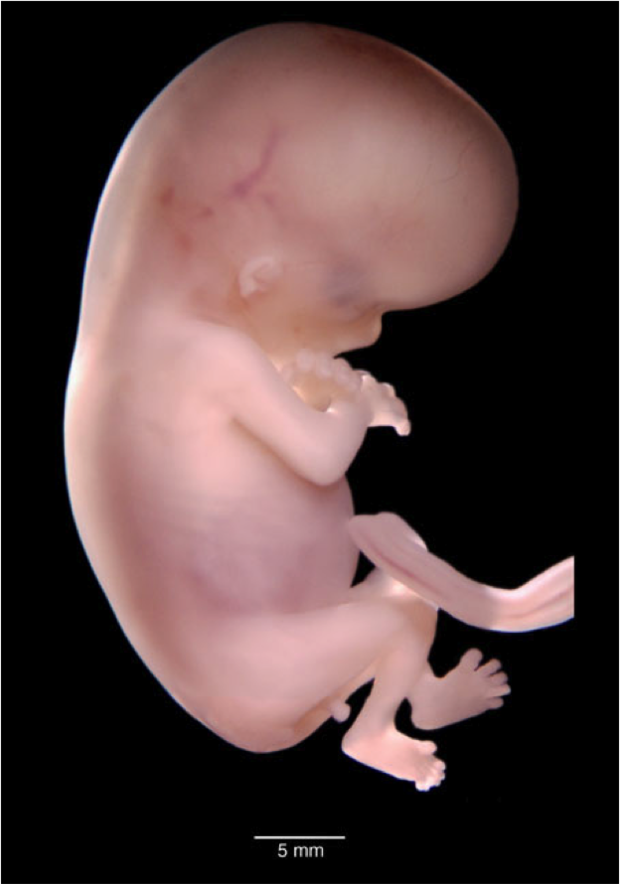 Its highest values in the case of the female fetus were noted in the spring. Testosterone production is significantly higher in pregnant women with male fetuses, mainly in the spring season. The stress resistance of pregnant women was higher in the summer months. Depending on the “fetal sex” factor, stress hormones in summer had the highest values in pregnant women with male fetuses. nine0021 Conclusions: The sex of the fetus is a significant factor influencing the seasonal variability in the production of melatonin, sex and stress hormones in pregnant women due to the formation of the specificity of systemic signaling between the maternal and fetal organisms in the dynamics of pregnancy.
Its highest values in the case of the female fetus were noted in the spring. Testosterone production is significantly higher in pregnant women with male fetuses, mainly in the spring season. The stress resistance of pregnant women was higher in the summer months. Depending on the “fetal sex” factor, stress hormones in summer had the highest values in pregnant women with male fetuses. nine0021 Conclusions: The sex of the fetus is a significant factor influencing the seasonal variability in the production of melatonin, sex and stress hormones in pregnant women due to the formation of the specificity of systemic signaling between the maternal and fetal organisms in the dynamics of pregnancy.
physiological pregnancy,
seasonal frequency,
hormonal status,
fetal sex
Botasheva T.L., Khloponina A.V., Vasilyeva V.V., Zavodnov O.P., Kaushanskaya L.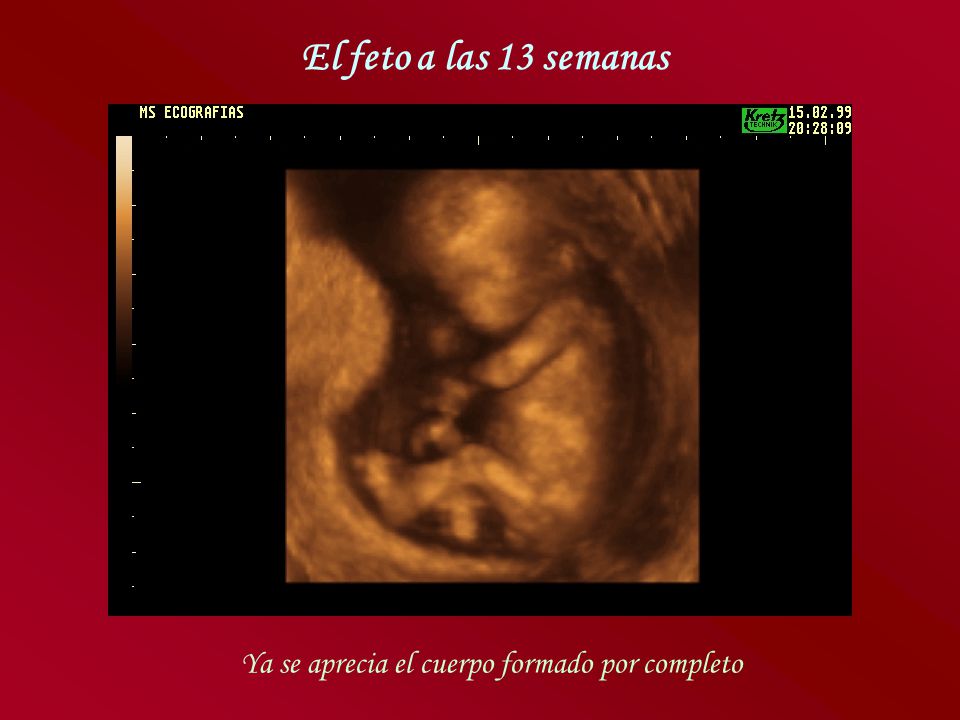 V., Zheleznyakova E.V. Seasonal periodicity of melatonin metabolism and hormonal status of pregnant women depending on the sex of the fetus. nine0039 Medical Bulletin of the South of Russia . 2018;9(3):70-76. https://doi.org/10.21886/2219-8075-2018-9-3-70-76
V., Zheleznyakova E.V. Seasonal periodicity of melatonin metabolism and hormonal status of pregnant women depending on the sex of the fetus. nine0039 Medical Bulletin of the South of Russia . 2018;9(3):70-76. https://doi.org/10.21886/2219-8075-2018-9-3-70-76
Botasheva T.L., Khloponina A.V., Vasil’eva V.V., Zavodnov O.P., Kaushanskaya L.V., Zheleznyakova E.V. Seasonal periodicity of melatonin exchange and hormonal status of pregnant women in dependence on fetus sex. nine0039 Medical Herald of the South of Russia . 2018;9(3):70-76.
(In Russ.)
https://doi.org/10.21886/2219-8075-2018-9-3-70-76
Currently, in general: a number of scientific works are increasingly using an interdisciplinary approach when considering obstetric problems. These include the chronophysiological aspect [1][2][3][4][5][6][7][8][9][10]. Previously, it was shown that gestational processes have a biorhythmic structure [3][11][12]. It was also found that the change in female fertility significantly depends on the seasons of the year [13][14][15]. A number of studies have noted that there is a relationship between the frequency of conception and seasonal rhythms of illumination [16][17][18][19], which consists in a positive correlation between the frequency of conception and the duration of the average annual illumination, as well as a negative correlation between the duration of the light period and the acrophase of the frequency of conception. In addition, it has been proven that, regardless of the season, childbirth occurs more often at night [11].
Previously, it was shown that gestational processes have a biorhythmic structure [3][11][12]. It was also found that the change in female fertility significantly depends on the seasons of the year [13][14][15]. A number of studies have noted that there is a relationship between the frequency of conception and seasonal rhythms of illumination [16][17][18][19], which consists in a positive correlation between the frequency of conception and the duration of the average annual illumination, as well as a negative correlation between the duration of the light period and the acrophase of the frequency of conception. In addition, it has been proven that, regardless of the season, childbirth occurs more often at night [11].
It has been shown that illumination, which changes at different times of the year and during the day, affects the activity of sexual behavior and the reproductive system of animals as a whole. As noted in the works of a number of authors [20][21], the pineal gland plays a major role both in the processes of photoperiodicity due to participation in the synthesis of hormones (melatonin and serotonin), and in changing the functional state of the gonads.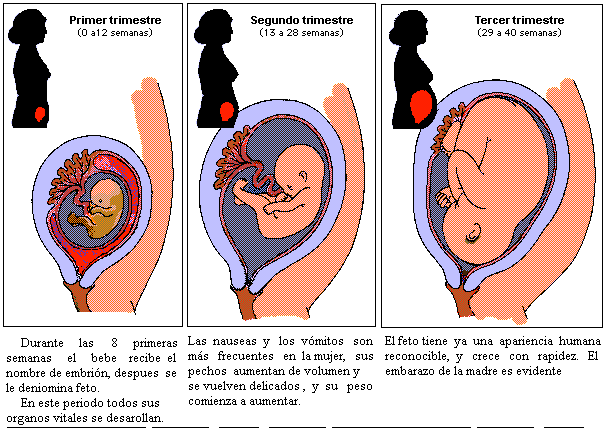 It is described that the pineal gland acts on the hypothalamic-pituitary system as an inhibitor [20][9]. As a result of a change in lighting, the rhythm of the formation of melatonin in the blood changes, which affects the state of the gonads and the reproductive system as a whole. It is noted that in humans and some animals during exposure to darkness, a decrease in the level of luteinizing hormone (LH) and follicle-stimulating hormone (FSH) in the peripheral blood and in the pituitary gland is noted when the activity of the pineal gland is high. It has been established that the content of prolactin, LH and FSH in blood plasma and in the pituitary gland is inversely related [3][22]. It is known that the synthesis of prolactin, carried out by acidophilic endocrinocytes of the adenohypophysis, is under the direct control of the hypothalamic structures, in particular, its supraoptic nuclei. At the same time, as a result of the rhythmic action of light, information is transmitted from the retina to the pineal gland along the retino-hypothalamic pathway, its activation or inhibition.
It is described that the pineal gland acts on the hypothalamic-pituitary system as an inhibitor [20][9]. As a result of a change in lighting, the rhythm of the formation of melatonin in the blood changes, which affects the state of the gonads and the reproductive system as a whole. It is noted that in humans and some animals during exposure to darkness, a decrease in the level of luteinizing hormone (LH) and follicle-stimulating hormone (FSH) in the peripheral blood and in the pituitary gland is noted when the activity of the pineal gland is high. It has been established that the content of prolactin, LH and FSH in blood plasma and in the pituitary gland is inversely related [3][22]. It is known that the synthesis of prolactin, carried out by acidophilic endocrinocytes of the adenohypophysis, is under the direct control of the hypothalamic structures, in particular, its supraoptic nuclei. At the same time, as a result of the rhythmic action of light, information is transmitted from the retina to the pineal gland along the retino-hypothalamic pathway, its activation or inhibition. Then, through the epiphyseal-hypothalamic system, due to hormonal emissions, the periodicity of functional processes in the reproductive system is ensured [3][23]. The activation of the pineal gland, which occurs at the onset of the dark time of the day, causes an increase in the production of melatonin and a decrease in serotonin [16][20][24]. As a result, there is an increase in the production of prostaglandins of the E2 class (PGE2) and a decrease in their level of the E2a class (PGE2a). At the same time, an increase in the level of melatonin with an increase in the PGE2 value is associated with inhibition of the contractile activity of the myometrium, which affects the nature of the course of labor [20]. A number of studies have found that, in addition to circadian rhythms, the female body is greatly influenced by annual and seasonal rhythms [15][25][3][26]. nine0005
Then, through the epiphyseal-hypothalamic system, due to hormonal emissions, the periodicity of functional processes in the reproductive system is ensured [3][23]. The activation of the pineal gland, which occurs at the onset of the dark time of the day, causes an increase in the production of melatonin and a decrease in serotonin [16][20][24]. As a result, there is an increase in the production of prostaglandins of the E2 class (PGE2) and a decrease in their level of the E2a class (PGE2a). At the same time, an increase in the level of melatonin with an increase in the PGE2 value is associated with inhibition of the contractile activity of the myometrium, which affects the nature of the course of labor [20]. A number of studies have found that, in addition to circadian rhythms, the female body is greatly influenced by annual and seasonal rhythms [15][25][3][26]. nine0005
In recent years, many works have appeared on the effect of the sex of the fetus on the nature of the course of gestational processes. The male sex of the fetus is recognized by the International Federation of Gynecologists and Obstetricians (FIGO) (2012) as a risk factor for the formation of threatening preterm birth [27][28]. There is evidence that during and after the hostilities, the number of male fetuses and newborns increased among the residents of the territories where they took place [16][29]. In the context of issues related to sexual dimorphism, the Trivers-Villard effect is of great importance, showing how the social and economic status of women affects the sex ratio of bearing children: women with a higher level of education have a higher sex ratio in favor of boys, and single women, those with very low incomes are more likely to have girls [19].
The male sex of the fetus is recognized by the International Federation of Gynecologists and Obstetricians (FIGO) (2012) as a risk factor for the formation of threatening preterm birth [27][28]. There is evidence that during and after the hostilities, the number of male fetuses and newborns increased among the residents of the territories where they took place [16][29]. In the context of issues related to sexual dimorphism, the Trivers-Villard effect is of great importance, showing how the social and economic status of women affects the sex ratio of bearing children: women with a higher level of education have a higher sex ratio in favor of boys, and single women, those with very low incomes are more likely to have girls [19].
Given the above, it seems relevant to study the characteristics of melatonin metabolism, as well as the hormonal status of women during gestation, depending on the sex of the fetus and seasonal biorhythms.
The purpose of the study was to identify seasonal features of melatonin metabolism and hormonal status in pregnant women depending on the sex of the fetus.
During the calendar year 2016, 1284 women with a physiological course of gestation at 37-40 weeks were examined, who were observed at the Rostov Research Institute of Obstetrics and Pediatrics under the Obstetric Monitoring program. Retrospectively, based on the results of hormonal and ultrasound studies, 286 patients with female fetuses (first group) and 254 pregnant women with male fetuses (second group) were selected according to the protocols of histories of observation of pregnant women. The randomization of the sample was carried out using the “Coin” method. nine0005
The study was conducted in accordance with the ethical standards of the Declaration of Helsinki and the Directives of the European Community (8/609EC).
Inclusion criteria were primigravida 2035 years with a singleton pregnancy. Excluded patients were multipregnant and multiparous, with multiple pregnancies, decompensated somatic morbidity, endocrine pathology, as well as with congenital malformations and chromosomal abnormalities in the fetus and after BPT programs (assisted reproductive technologies). nine0005
nine0005
As part of the study, the sex of the fetus was determined using ultrasound scanning (Voluson E8 Expert (Austria)). Assessment of the hormonal status included the determination of the level of cortisol, ACTH, progesterone, testosterone, estradiol, in the venous blood and 6-sulfate-oxy-melatonin in the urine of pregnant women (morning portion). For the quantitative determination of the level of hormones in the blood, the method of enzyme immunoassay and various test systems were used: “Steroid ELISA-cortisol-01” (Russia) to determine the level of cortisol; “ASTN ELISA” (USA) to determine the level of ACTH; “SteroidIFA-17-OH-progesterone” (Russia) to determine the level of progesterone; “DRG INSTRUMENTS, G.M.B.H.” (Germany) to determine the level of testosterone; “uE3 kit”, (Finland) for the study of estradiol production. The determination of melatonin in the urine of women was carried out by enzyme immunoassay (ELISA test system, Germany) by assessing the level of its main metabolite 6-sulfatoxymelatonin, using the computer program “Viktor-Wallak”, Finland. Urine and blood sampling for research was carried out before the course of vitamin therapy. nine0005
Urine and blood sampling for research was carried out before the course of vitamin therapy. nine0005
Statistical analysis of the results was carried out by parametric methods. The data obtained are presented as relative values (%), as well as M ± m. To assess the difference in arithmetic means, Student’s t-test was used. Differences between the parameters were assessed using Fisher’s F-test. The Shapiro-Wilk test was used to check the normality of distribution in the samples. For multiple comparisons, the Bonferoni correction was introduced. Null hypothesis testing was carried out using the t, F and X criteria 2 , at p<0.05 significance level. Statistical processing of the results was carried out using the application packages Statistica 6.0 for Windows.
During the analysis of the obtained data, it was found that in all pregnant women, regardless of the sex of the fetus, the lowest level of melatonin was determined in the so-called light-saturated periods of the year – in the summer and spring months (Table 1). The highest levels of melatonin were observed in all patients, mainly in winter. At the same time, there were no significant differences in melatonin levels between the selected groups within the season. nine0005
The highest levels of melatonin were observed in all patients, mainly in winter. At the same time, there were no significant differences in melatonin levels between the selected groups within the season. nine0005
The level of hormones in pregnant women of clinical groups depending on the seasons of the year
|
Indicators Indicators |
Spring Spring |
Summer Summer |
||
|---|---|---|---|---|
|
Pregnant women with fetuses Pregnant women with female fetuses |
Pregnant women with male fetuses Pregnant with male fetuses |
Pregnant women with fetuses female Pregnant women with female fetuses |
Pregnant women with male fetuses Pregnant with male fetuses |
|
|
Melatonin ng/ml Melatoninng/ml |
178.2±16. |
161.6±12.9* |
168.2±11.3* |
152.5±10.5* |
|
Cortisol nmol/ml Cortisol nmol/ml |
958.3±27.6*/** |
1220.6±23.5*/** |
571.7±29.9 */** |
813.1±24.3*/** |
|
ACTH pg/ml ACTHpg / ml |
nine0010 20.3±1.2*/** |
29.7±1.9 */** |
9.3±1.2*/** |
19.6±1.3*/** |
|
Progesterone nmol/l Progesterone nmol /1 |
821.7±11.7*/** |
539.2±12.9*/** |
749.5±8.2 */** |
563.2±12.1*/** |
|
Estradiol pmol/l Estradiol pmol /1 |
57.244.5* |
49.3±2.3* |
67.1±2.2* |
50.0±2. |
|
Testosterone nmol/l Testosterone nmol /1 |
2.95±0.8* |
7.08±0.4*/** |
2.82±1.4* |
4.85±1.3* |
|
Autumn Autumn |
Winter Winter |
|||
|
Melatonin ng/ml Melatoninng/ml |
229.4 ±10.8* |
187.1±10.1* |
238.5 ±9.4*/** |
198.2±12.1 /** |
|
Cortisol nmol/ml Cortisol nmol/ml |
929.9±25.3 */** |
nine0010 1304.2±1.9*/** |
1429.1±21.4*/** |
1296.4±23.5*/** |
|
ACTH pg/ml ACTGpg/ml |
27.3±1.5*/** |
36.8±1.9*/** |
36.4±2. |
38.1±1.4*/** |
|
Progesterone nmol/l Progesterone nmol /1 |
454.2±14.7*/** |
294.8±9.6 */** |
507.6±18.1*/** |
422.1±16.4*/** |
|
Estradiol pmol/l Estradiol pmol /1 |
52.1±3.2* |
50.2±2.5* |
54.5±3.8*/** |
52.0±1.9* |
|
Testosterone nmol/l Testosterone nmol /1 |
4.03±1.7* |
2.41±1.1* |
4.03±1.5* |
|
(p<0.05) * — statistical significance of differences in similar parameters in patients of different groups; ** - statistical significance of differences in the same indicators within the group, depending on the season of the year.
Note, (p <0. 05) * — the statistical significance of the differences in the same indicators in patients of different groups; ** — significant statistical of the differences of the same indices within the group depending on the season of the year. nine0005
05) * — the statistical significance of the differences in the same indicators in patients of different groups; ** — significant statistical of the differences of the same indices within the group depending on the season of the year. nine0005
Changing the level of illumination in the dynamics of different seasons of the year and the subsequent change in melatonin production significantly affect the functional state of the reproductive system and the secretion of sex hormones. In the process of analyzing the indicators of estradiol and progesterone, it was found that the women of the first group had significantly higher rates than those of the second group, regardless of the time of year. In pregnant women with female fetuses, the highest values of these indicators were recorded mainly in the summer. In patients of the second group, there were no significant differences in estradiol values in different seasons. The data obtained also reflect the maximum values of progesterone for women of the first group in the spring and summer months of the year, and its lowest level in the autumn period when daylight hours are shortened.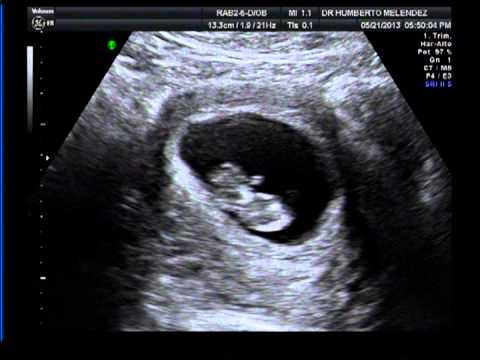 nine0005
nine0005
The value of testosterone was significantly higher in women of the second group in all seasons. The highest values were determined in spring (against the background of an increase in the daylight period). In women of the first group, there were no seasonal differences in the level of this hormone.
The study showed that in pregnant women of the first group, the values of cortisol and ACTH in all seasons were significantly lower than in patients of the second group. When analyzing the level of these hormones, it was also found that the highest values were observed in women of the first group in winter, and in the second group – in autumn and winter, while the lowest values in patients of both groups were recorded in summer. nine0005
The obtained results indicate that the sex of the fetus is a significant factor influencing the seasonal variability in the production of melatonin, sex and stress hormones in pregnant women due to the formation of specific system signaling between the maternal and fetal organisms. An increase in the level of melatonin, mainly in the autumn and winter lightless seasons, indicates an increase in the function of the pineal gland under conditions of seasonal light deprivation. In the light-saturated spring season, there is an activation of subsystems that mediate sexual function and an increase in the production of sex hormones. In winter, an increase in the level of ACTH and cortisol is recorded, due to a more pronounced activity of stress-liberating subsystems. In all cases of seasonal variability of hormonal status in pregnant women, the leading role of light and temperature factors, characteristic of different seasons of the year, in the regulation of the activity of the hormonal system can be traced. nine0005
An increase in the level of melatonin, mainly in the autumn and winter lightless seasons, indicates an increase in the function of the pineal gland under conditions of seasonal light deprivation. In the light-saturated spring season, there is an activation of subsystems that mediate sexual function and an increase in the production of sex hormones. In winter, an increase in the level of ACTH and cortisol is recorded, due to a more pronounced activity of stress-liberating subsystems. In all cases of seasonal variability of hormonal status in pregnant women, the leading role of light and temperature factors, characteristic of different seasons of the year, in the regulation of the activity of the hormonal system can be traced. nine0005
The diversity of seasonal representation of hormone production in mothers bearing fetuses of the opposite sex indicates the existence of differences in functional processes in the system “mother-placenta-fetus male” and “mother-placenta-fetus female”, due to the specifics of seasonal biorhythms of the hormonal and nervous regulation responsible for the transfer of information from the mother to the fetus and, conversely, in the case of a male or female fetus, the possibility of a “training” influence of the seasonal periodicity of the hormonal function in the maternal organism on the fetal organism is not excluded, which can determine the seasonal specificity of physiological processes in children at the postpartum stage ontogeny. nine0005
nine0005
 Depending on the “fetal sex” factor, stress hormones in summer had the highest values in pregnant women with male fetuses.
Depending on the “fetal sex” factor, stress hormones in summer had the highest values in pregnant women with male fetuses. 1. Romanov Yu.A. biological rhythms. In book. Problems of space biorhythmology. – M.: Nauka; 1980.
2. Arushanyan E.B. Modern aspects of chronophysiology and chronopharmacology. – Stavropol: StGMA; 2004.
3. Agadzhanyan N.A., Petrov V.I., Radysh I.V., Krayushkin S.I. Chronophysiology, chronopharmacology and chronotherapy: Monograph. Volgograd: VolGMU Publishing House; 2005.
4. Golikov A.P., Golikov P.P. Seasonal biorhythms in physiology and pathology. – M.: Medicine, 1973.
5. Komarov F.I., Zakharov L.V., Lisovsky V.A. Daily rhythm of physiological functions in a healthy and sick person. – L.: Medicine, 1966.
6. Zaslavskaya R.M., Vaskova L.B., Bolsunovskaya Yu.R. Chronopharmacology and chronomedicine as a new methodological approach to optimizing treatment. //Space and time. – 2012. – No. 1 (7). – S. 195-198.
1 (7). – S. 195-198.
7. Khetagurova L.G., Rapoport S.I., Botoeva N.K. Stages of formation of chronobiology and chronomedicine in Russia (historical essay). // Space and time. – 2013. – No. 2 (12). – p. 229-237.
8. Botasheva T.L., Rogova N.A., Chernositov A.V., Kaushanskaya L.V., Shubitidze M.G. Seasonal biorhythms of the functional system “mother-placenta-fetus” depending on its stereofunctional organization in physiological and complicated pregnancy. // Tauride Medical and Biological Bulletin. – 2013. – T. 16. – No. 2-1 (62). – S. 32-35.
9. Kovalzon V.M., Dorokhov V.B. The cycle of wakefulness – sleep and human biorhythms in various modes of alternating light and dark periods of the day. // Health and education in the XXI century. – 2013. – T. 15. – No. 1-4. – S. 151-162. nine0005
10. Strizhakov A.N., Tezikov Yu.V., Lipatov I.S., Martynova N.V., Zhernakova E.V., et al. » in physiological and complicated pregnancy. // Issues of gynecology, obstetrics and perinatology.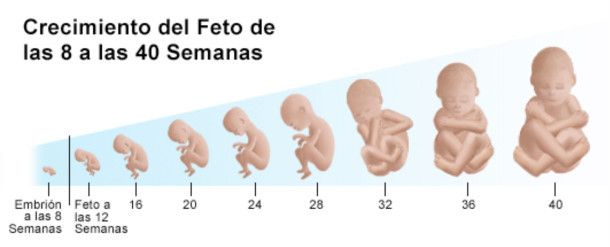 – 2017. – T. 16. – No. 1. – P. 25-32. doi: 10.20953/1726-1678-2017-1-25-32
– 2017. – T. 16. – No. 1. – P. 25-32. doi: 10.20953/1726-1678-2017-1-25-32
11. Rogova N.A., Botasheva T.L., Frolov A.A., Kapustin E.A., Chernositov A.V., Palieva N.V. Adaptation status of pregnant women in the prenatal period depending on the stereoisomerism of the uteroplacental complex and daily photoperiodism in different seasons of the year. // Modern problems of science and education. – 2013. – No. 5. – P. 286.
12. Arushanyan E.B., Baturin V.A., Ovanesov K.B. Fundamentals of chronomedicine and chronopharmacology. – Stavropol; 2016.
13. Romanov Yu.A. Problems of chronobiology.– M.: Knowledge; 1989.
14. Ashoff Yu. Biological rhythms. In two volumes. – M .: Mir; 1984.
15. Radysh I.V. Circadian rhythm in women. In: Health of the population and physical education. – Cheboksary, 1994.
16. Geodakyan S.V. Two genders. Why and why? Evolutionary theory of sex. – Moscow; 2011.
17. Geodakyan V.A. Evolutionary logic of sex differentiation and longevity.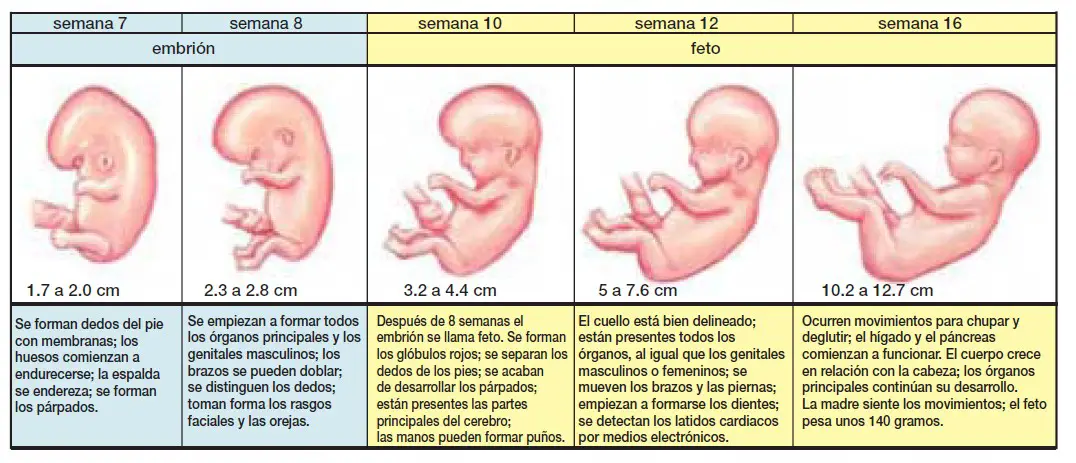 // Nature. – 1983. – No. 1. – P. 70-80.
// Nature. – 1983. – No. 1. – P. 70-80.
18. Rapoport S.I. Chronomedicine, circadian rhythms. Who needs it? // Clinical medicine. – 2012. – T. 90. – No. 8. – S. 73-75.
19. Zhang WX, Chen SY, Liu C. Regulation of reproduction by the circadian rhythms. // Acta Physiol Sinica. – 2016. – V. 68(6). – R. 799-808.
20. Anisimov V.N. Pineal gland, biorhythms and aging of the organism // Advances in physiological sciences. – 2008. – T. 39. – No. 4. – S. 40-65.
21. Gubin D.G. Molecular mechanisms of circadian rhythms and principles of development of desynchronosis. // Successes of physiological sciences. – 2013. – T. 44. – No. 4. – S. 65-87.
22. Serov V.N., Prilepskaya V.N., Ovsyannikova T.V. Gynecological endocrinology. Management. – M.: MED-press-inform; 2015.
23. Chronobiology and chronomedicine: Guide. Ed. S.I. Rapoport, V.A. Frolova, L.G. Khetagurova. – M .: LLC “Medical Information Agency”; 2012.
24. Botasheva T.L., Linde V.A., Sargsyan O.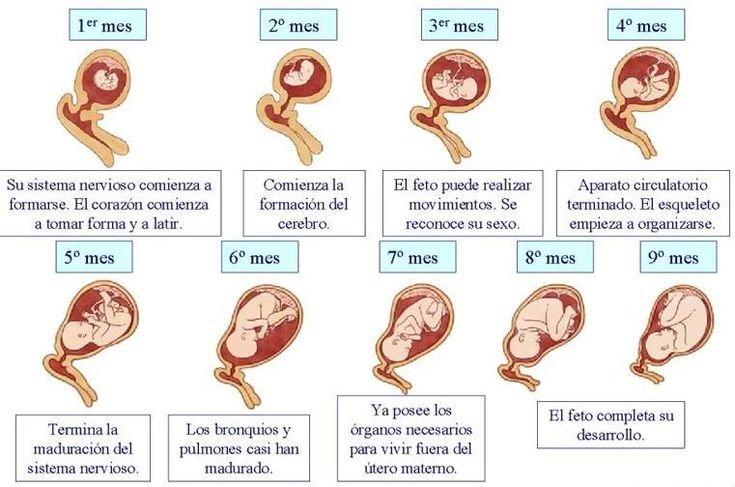 D., Ermolova N.V., Gimbut V.S., et al. cytokines in women with an alternative sex of the fetus. // Modern problems of science and education. – 2014. – No. 6. – P. 1057.
D., Ermolova N.V., Gimbut V.S., et al. cytokines in women with an alternative sex of the fetus. // Modern problems of science and education. – 2014. – No. 6. – P. 1057.
25. Komarov F.I., Rapoport S.I. Chronobiology and chronomedicine.– M.: Triada-X; 2000.
26. Volobuev A.N., Pyatin V.F., Romanchuk N.P. Circadian biophysics and chronomedicine. // Health and education in the XXI century. – 2016. – T. 18. – No. 5. – P. 97-100.
27. Di Renzo GC, Roura LC, Facchinetti F, Helmer H, Hubinont C, Jacobsson B. et al. Preterm Labor and Birth Management: Recommendations from the European Association of Perinatal Medicine // Journal of Maternal-Fetal and Neonatal Medicine. – 2017. – V. 30(17). – R. 2011-2030. doi: 10.1080/14767058.2017.1323860
28. Hesketh T, Xing ZW. Abnormal sex ratios in human populations: Causes and consequences. // Proc Natl Acad Sci USA. – 2006. – V. 103(36). – R. 13271-13275. doi: 10.1073/pnas.0602203103. nine0005
29. Chacon-Puignau GC, Jaffe K.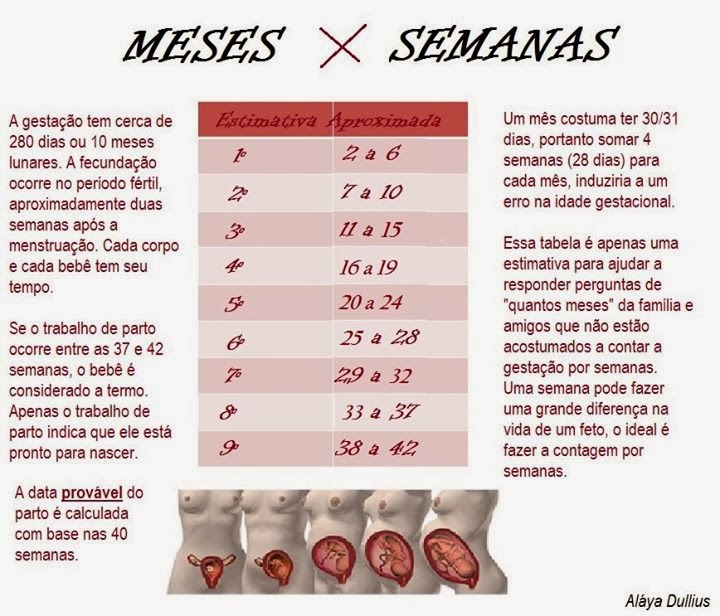 Sex ratio at birth deviations in modern Venezuela: the Trivers-Willard effect. // Soc Biol. – 1996. – V. 43. – R. 257-270.
Sex ratio at birth deviations in modern Venezuela: the Trivers-Willard effect. // Soc Biol. – 1996. – V. 43. – R. 257-270.
T. L. Botasheva
Rostov State Medical University, Rostov-on-Don
Russia
Botasheva Tatyana Leonidovna, Doctor of Medical Sciences, Professor, Chief Researcher of the Department of Medical and Biological Problems in Obstetrics, Gynecology and Pediatrics
eLibrary SPIN: 3341-2928
Conflict of Interest: No conflict of interest.
A. V. Khloponina
Rostov State Medical University, Rostov-on-Don
Russia
Anna Valerievna Khloponina, Candidate of Medical Sciences, Senior Researcher, Obstetrics and Gynecology Department
Conflict of Interest: There is no conflict of interest.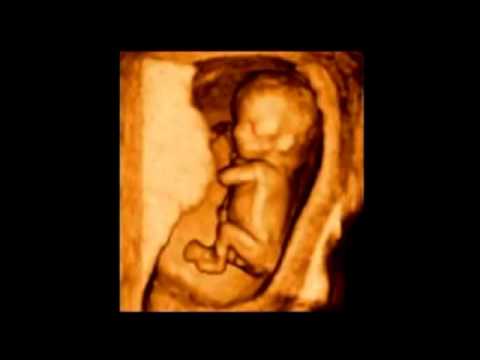
nine0003 V. V. Vasilyeva
Rostov State Medical University, Rostov-on-Don
Russia
Vasilieva Valentina Valerievna, Doctor of Biological Sciences, Associate Professor, Leading Researcher, Department of Biomedical Problems in Obstetrics, Gynecology and Pediatrics
Conflict of Interest: No conflict of interest.
O. P. Zavodnov
Rostov State Medical University, Rostov-on-Don
nine0034 Russia
Zavodnov Oleg Pavlovich, PhD, Researcher, Department of Biomedical Problems in Obstetrics, Gynecology and Pediatrics
SPIN code: 7234-3875
Conflict of interest: No conflict of interest.
L. V. Kaushanskaya
Rostov State Medical University, Rostov-on-Don
Russia
Kaushanskaya Lyudmila Vladimirovna, Doctor of Medical Sciences, Associate Professor, Chief Researcher of the Obstetrics and Gynecology Department
nine0034 Conflict of interest: No conflict of interest.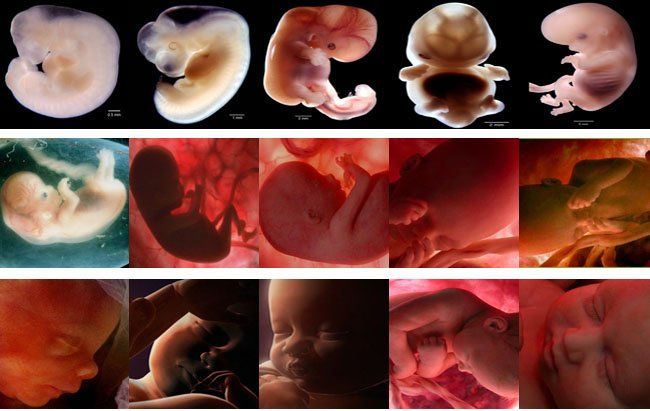
E. V. Zheleznyakova
Rostov State Medical University, Rostov-on-Don
Russia
Elena Vasilievna Zheleznyakova, Candidate of Medical Sciences, Researcher, Department of Biomedical Problems in Obstetrics, Gynecology and Pediatrics
SPIN code: 5805-3908
Conflict of Interest: No conflict of interest. nine0005
Botasheva T.L., Khloponina A.V., Vasilyeva V.V., Zavodnov O.P., Kaushanskaya L.V., Zheleznyakova E.V. Seasonal periodicity of melatonin metabolism and hormonal status of pregnant women depending on the sex of the fetus. nine0039 Medical Bulletin of the South of Russia . 2018;9(3):70-76. https://doi.org/10.21886/2219-8075-2018-9-3-70-76
Botasheva T.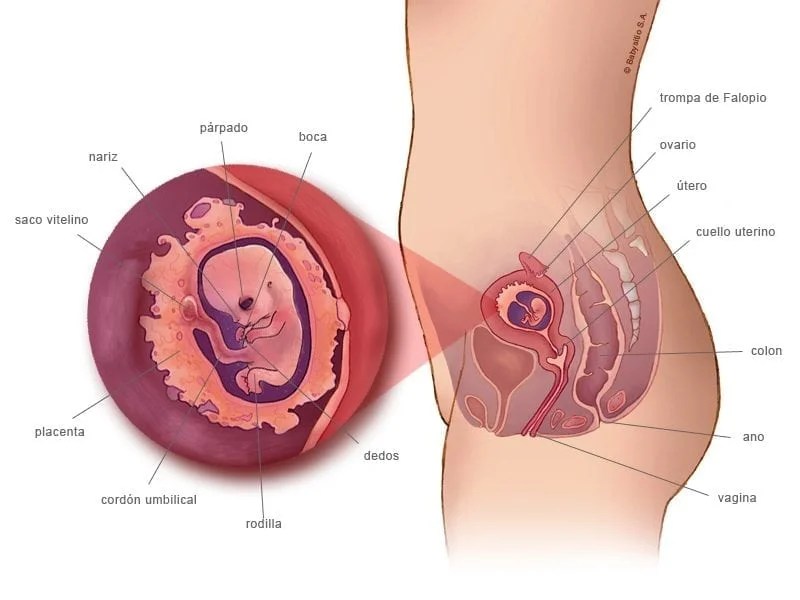 L., Khloponina A.V., Vasil’eva V.V., Zavodnov O.P., Kaushanskaya L.V., Zheleznyakova E.V. Seasonal periodicity of melatonin exchange and hormonal status of pregnant women in dependence on fetus sex. nine0039 Medical Herald of the South of Russia . 2018;9(3):70-76.
L., Khloponina A.V., Vasil’eva V.V., Zavodnov O.P., Kaushanskaya L.V., Zheleznyakova E.V. Seasonal periodicity of melatonin exchange and hormonal status of pregnant women in dependence on fetus sex. nine0039 Medical Herald of the South of Russia . 2018;9(3):70-76.
(In Russ.)
https://doi.org/10.21886/2219-8075-2018-9-3-70-76
Views: 536
Content is available under the Creative Commons Attribution 4.0 License.
Dakar is the capital and largest port of Senegal. Despite the fact that the city is large, it is very poor. Among its main attractions are the African Renaissance Monument (the tallest statue in Africa) and the Dakar Cathedral Mosque.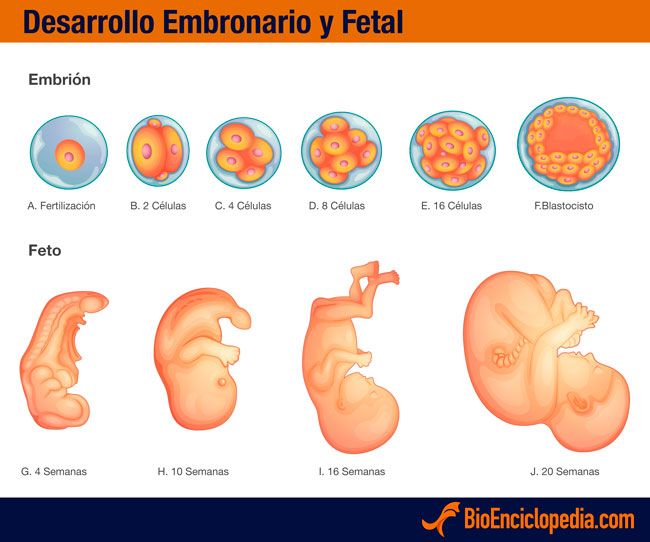 It will be interesting for a tourist to watch the fights of local wrestlers on the sites of the city, in front of which they perform unusual ritual dances and ablutions. In the vicinity of Dakar, you can visit the pink lake Retba. It impresses with its unusual, rich color.
It will be interesting for a tourist to watch the fights of local wrestlers on the sites of the city, in front of which they perform unusual ritual dances and ablutions. In the vicinity of Dakar, you can visit the pink lake Retba. It impresses with its unusual, rich color.
Another unusual place is Acro Baobab, the only rope park in the world organized on baobabs. Just 10 km from the capital is the resort town of Les Almadie. Its rocky coastline attracts divers and snorkelers.
nine0005
St. Louis. The ancient capital of Senegal, located on a narrow island of the river of the same name. Here the Europeans once founded the first settlement. The city quickly filled with rich French people who competed in their wealth by building luxurious mansions. Louis Historic Center is now a UNESCO World Heritage Site. Just 20 km south of the former colonial capital, there is the beautiful Lang de Barbary National Park with graceful pink flamingos. And 60 km to the north is the third largest ornithological park in the world – Birds of Dzhudzh. The old city is famous not only for historical and natural sights. Every year in May, a jazz festival is held here – the largest on the continent, and in January-February, blues fans come here for their holiday. nine0005
The old city is famous not only for historical and natural sights. Every year in May, a jazz festival is held here – the largest on the continent, and in January-February, blues fans come here for their holiday. nine0005
Kaolak is the peanut capital, where peanut plantations spread around the city. Among the sights, it is worth noting the most beautiful Moroccan-style mosque in the country (entrance inside is closed for non-Muslims). In the vicinity of the city, tourists are attracted by the Salum River Delta, where everything is covered with mangroves, and oysters grow in salt water, on the roots of trees. It should be noted that Kaolak is one of the dirtiest cities in Africa, so outbreaks of epidemics of malaria, cholera, yellow fever and even leprosy are regularly recorded here. nine0005
Ziguinchor is a city that still has architecture that tells of its colonial past. And the fact that this is a real African city is reminiscent of one of the best markets in Senegal and aboriginal villages in the vicinity.