Embarazo
La primera foto de tu hijo es, sin duda, la ecografía. Por medio de este examen podrás ver, escuchar y sentir a tu guagua, sus movimientos acuáticos y el latido de su corazón. ¿Cómo va creciendo el embarazo semana a semana?
Embarazo de 1 semana:
En la primera semana de embarazo el óvulo fecundado desciende por las trompas de Falopio hasta llegar al útero. Este será su refugio durante los próximos 9 meses. En esta etapa puede que aún no te enteres de que estás acompañada, pero pronto empezarán las señales.
Embarazo de 3 semanas:
¿Tienes la sospecha de que estás embarazada? Si es así, el óvulo fecundado ya está creciendo e instalándose en la pared de tu útero. A pesar del poco tiempo, el corazón de tu hijo es un grupo de células musculares y diminutas que ya se está formando.
Embarazo de 4 semanas:
Tu hijo (a) está en su período inicial de embriogénesis que va desde el óvulo fecundado que se divide en múltiples células, las que se van especializando en formar cada tejido del cuerpo. Es un ciclo muy importante porque es aquí es donde ocurre la mayoría de los problemas -muchas veces graves- en el desarrollo que lleven a una malformación.
Embarazo de 5 semanas:
Ecográficamente es posible reconocer en el interior del útero, el saco gestacional, mientras que su tamaño es casi como una semilla de sésamo.
En esta instancia está formado por células que, más tarde, formarán sus tejidos y órganos. De hecho, su sistema circulatorio ya se está formando.
Por otro lado, a las 5 semanas, la placenta y el cordón umbilical ya están empezando a funcionar, llevando oxígeno y nutrientes a tu hijo.
Embarazo de 6 semanas:
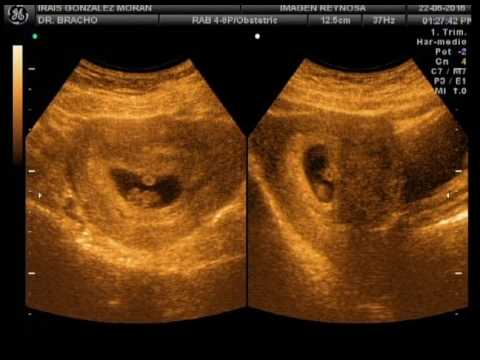
En este período ya existe un embrión que mide de 2 a 3 milímetros.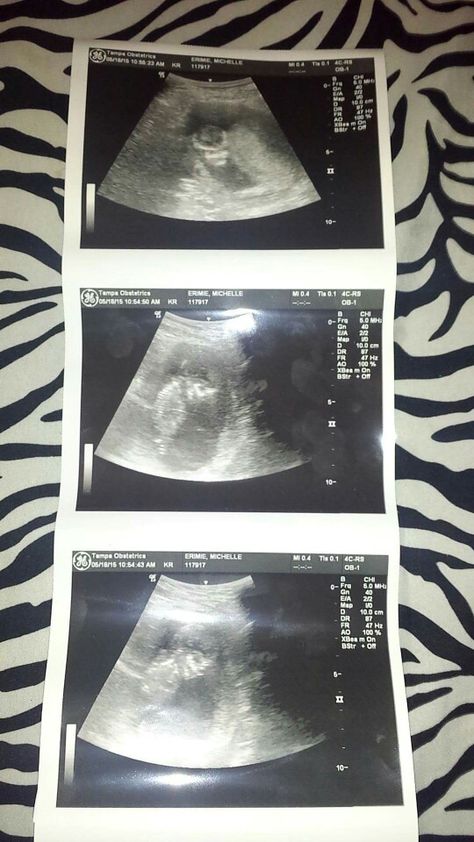 Tiene cabeza y un corazón que late, con un sistema circulatorio muy primario. Es posible reconocer una pequeña vesícula (saco vitelino) en la cual el embrión fabrica los primeros glóbulos rojos.
Tiene cabeza y un corazón que late, con un sistema circulatorio muy primario. Es posible reconocer una pequeña vesícula (saco vitelino) en la cual el embrión fabrica los primeros glóbulos rojos.
La visualización del embrión ya es posible desde el final de la semana 5, o el inicio de la semana 6, y los latidos del corazón visibles en la ecografía son habitualmente después de la semana 6.
Embarazo de 7 semanas:
Antes de las séptima semanas puede que se vea el saco gestacional sin un embrión en su interior. No te asustes, ya que puede que sea aún muy pequeño para ser visto. ¡Paciencia!
Embarazo de 8 semanas:
Entre las sexta y las décima semanas se forman esencialmente todas las estructuras internas y externas del adulto. A las 8 semanas, el embrión mide 20 milímetros. Empiezan a definirse mejor las facciones humanas. La cabeza aparece muy grande en relación con el cuerpo. Los ojos están cubiertos por una piel intacta que se abrirá posteriormente para transformarse en los párpados. Tiene brazos y piernas, aunque las manos y los pies aún no están completamente formados. El feto ya se mueve, aunque no pueda sentirlo.
Tiene brazos y piernas, aunque las manos y los pies aún no están completamente formados. El feto ya se mueve, aunque no pueda sentirlo.
Embarazo de 9 semanas:
A esta altura comienzan a aparecer las rodillas y los codos, mientras que sus costillas ya se están desarrollando para cubrir sus delicados órganos internos.
Si en la primera ecografía viste una cola, en esta etapa comienza a desaparecer. Además, su sistema nervioso ya se está conectando con todo el cuerpo, realizando pequeños movimientos, imperceptibles para tu.
Embarazo de 11 semanas:
Entre la semana 11 y 14 se realiza el segundo examen, generalmente por vía abdominal, entre las 11 y 14 semanas de embarazo. Esto, con el fin de evaluar la anatomía fetal y descartar anormalidades estructurales importantes.
También se puede detectar el riesgo de algunas anomalías cromosómicas mediante la medición del espesor del tejido de la región nucal o translusencia nucal, la presencia del hueso nasal, el flujo en el ductus venoso, entre otros. Durante este examen se puede llegar a conocer el género, el que se confirma tras la semana 18 a 20 o quinto mes.
Durante este examen se puede llegar a conocer el género, el que se confirma tras la semana 18 a 20 o quinto mes.
Embarazo de 12 semanas:
El feto es fácilmente reconocible como ser humano, con una cabeza aún grande, manos y pies formados y brazos proporcionados para su cuerpo. Se pueden ver las costillas y las vértebras de la columna.
Embarazo de 13 semanas:
Tu hijo (a) ya tiene uñas y ya se mueve vigorosamente. Cierra los puños y puede abrir la boca. Se han formato todos los órganos internos y algunos ya funcionan. Tiene algunos reflejos, por lo que se mueve si tocas tu vientre, sin embargo, aún no podrás sentirlo. Mide aproximadamente 7 cm.
Embarazo de 15 semanas:
Tu hijo (a) ya mide unos 10 cm de cabeza a cola y se moverá libremente en la cavidad uterina. A esta altura sus órganos ya están formados y queda completar su desarrollo en crecimiento y maduración funcional hasta completar el embarazo.
Embarazo de 16 semanas:
Tu hijo (a) sigue creciendo. La piel se ha engrosado y se cubre de un fino vello llamado lanugo. Aparecen las cejas y las pestañas y ya está midiendo entre 21 y 22 cm.
La piel se ha engrosado y se cubre de un fino vello llamado lanugo. Aparecen las cejas y las pestañas y ya está midiendo entre 21 y 22 cm.
Embarazo de 18 semanas:
A esta altura del embarazo ya puedes confirmar el sexo de tu guagua, sin embargo, es probable que tu médico prefiera esperar un poco más. ¡Falta poco!
Embarazo de 20 semanas
Sus movimientos son cada vez más fuertes y la mamá ya los puede sentir. Comienza a crecer pelo en su cabeza y los ojos son prominentes, ya que aún no se ha redondeado la cara.
Parece un poco delgado, pero comienza a depositarse grasa bajo su piel, lo que le dará un aspecto de niño sano.
Embarazo de 22 semanas
Al final de este período (22-24 semanas) ocurre habitualmente el segundo importante control donde se puede confirmar el sexo (esto puede ser en realidad desde la semana 16-18), verificando que su anatomía esté bien y, así, realizar algunas predicciones de riesgo de problemas en el embarazo.
Embarazo de 23 semanas
La piel se cubre de una sustancia grasa protectora llamada unto sebáceo que protege la piel dentro del útero y que se une al pelo.
Embarazo de 24 semanas
Ya puede abrir sus ojos y escuchar los sonidos. Las palmas de las manos y las plantas de los pies tienen surcos reconocibles e, incluso, huellas digitales. Los órganos detales están bien constituidos y de un tamaño adecuado para que puedan ser explorados con detalle por el ecografista.
Al final de este período (24 semanas) puede pesar un promedio de 750 gramos.
Embarazo de 25 semanas
A esta altura tu hijo (a) mide 20 cm aproximadamente y se cree ya escucha. Algunos papás inician un plan de estimulación con música, o simplemente les habla. Inténtalo.
Embarazo de 26 semanas
A esta altura tienes que programar el siguiente examen. Aquí se estudia en detalle la anatomía fetal y la irrigación uterina placentaria. Pueden ser diagnosticadas la mayor parte de las alteraciones anatómicas severas, como también crecimiento fetal, características de la placenta, entre otros.
Pueden ser diagnosticadas la mayor parte de las alteraciones anatómicas severas, como también crecimiento fetal, características de la placenta, entre otros.
Gracias a este examen, también se puede determinar el riesgo de un parto prematuro, mediante la medición del cuello uterino por vía transvaginal.
Embarazo de 27 semanas
Tu guagua se sigue desarrollando en maduración de órganos, especialmente el sistema nervioso. Tu ginecólogo vigilará que el crecimiento fetal esté de acuerdo a los esperado.
Embarazo de 28 semanas
El feto crece y crece. Sus movimientos son más frecuentes y visibles, especialmente, cuando estás acostada.
Pesa aproximadamente entre 1.000 y 1.200 gramos y mide 35 centímetros. En los siguientes meses, el feto crecerá, ganará peso y madurará en forma importante, creándose las mejores condiciones para adaptarse a la vida extrauterina.
Embarazo de 32 semanas
El niño engorda a sus anchas. Pesa entre 1.800 y 2.000 gramos y mide entre 40 y 42 centímetros.
Pesa entre 1.800 y 2.000 gramos y mide entre 40 y 42 centímetros.
La piel se engruesa por el depósito de grasa que le servirá de aislante al nacer. Puede que comience a seguir pequeñas contracciones del útero, no te alarmes, tu hijo se prepara para el gran día.
Aquí se realiza el cuarto examen para ver el crecimiento, talla y vitalidad de tu hijo. Además, ya es posible ver la proyección de peso al término del embarazo.
Embarazo de 33 semanas
Tu hijo (a) cada día está más desarrollado y con piel más gruesa. A las 32-34 semanas pesa cerca de 2 a 2,4 kilos, por lo que -de nacer- tiene altas probabilidades que todo salga bien… pero aún es mejor esperar.
Embarazo de 34 semanas
Tu hijo (a) se está acomodando y puede que busque la posición denominada “cefálica”. Esto es normal y no quiere decir que vaya a nacer antes de tiempo.
Embarazo de 35 semanas:
Es posible que inicie sus primeras incursiones en la pelvis buscando el canal del parto, lo que genera algunas bruscas sensaciones de compresión rectal (ganas de ir al baño) y vesicales. Si aún está con la cabeza hacia arriba no te preocupes, algunas guaguas se dan vuelta al final del embarazo.
Si aún está con la cabeza hacia arriba no te preocupes, algunas guaguas se dan vuelta al final del embarazo.
Embarazo de 36 semanas
En este período se produce un incremento del peso y se completa el proceso de maduración pulmonar. La cabeza tiende a bajar hacia la pelvis y los movimientos disminuyen, pero nunca desaparecen (ya es muy grande y le cuesta más moverse dentro del útero).
Embarazo de 37 semanas
Cada día tu hijo (a) estará más maduro, en su piel, cerebro y pulmones. De nacer en forma espontánea, lo más probable es que sea perfecto. Asegúrate de tener un bolso listo en caso de tener que partir rumbo al parto.
Embarazo de 38 semanas
Comenzó la cuenta regresiva para el parto. Tu hijo(a) comienza a deshacerse de la capa de grasa que lo recubre y permanece en suspensión en el líquido amniótico, mientras que sus pulmones ya se están preparando para ese día. En esta etapa la placenta reduce la emisión de progesterona y fomenta la producción de oxitocina con el fin de regular las contracciones del útero y la dilatación.
Embarazo de 39 semanas
Ha crecido mucho durante los últimos días y seguramente se siente muy apretado, sin embargo, se mantendrá activo hasta el momento de parto. Sus órganos ya están desarrollados, pues ya se considera un embarazo de término. A esta altura el lanugo ha desaparecido en gran parte y sus uñas siguen creciendo. Por otra parte, sigue percibiendo estímulos desde el exterior, así que sigue hablándole y cantándole si prefieres.
Si eres madre primeriza y aún no sabes cuáles son las señales de parto, aquí te las indicamos.
Embarazo de 40 semanas
Son pocos los partos que llegan a fin. Si aún estás en esta semana, seguro te sientes muy cansada, pero ánimo porque falta muy poco. Tu hijo(a) ya se desarrolló y está en perfectas condiciones para vivir fuera del útero.
Vista 438347 veces
Una de las pruebas por las que inevitablemente ha de pasar una embarazada es la ecografía. Serán necesarias al menos tres ecografías durante todo el embarazo, una por cada trimestre del embarazo. Pero es la primera ecografía la que más expectación genera, la que más ilusión hace, pero también la que más nervios produce en la mamá. ¿Estás preparada para tu primera ecografía? En Guiainfantil.com te contamos cuándo se hace, para qué sirve, qué verás en ella, etc. ¡Felicidades por tu bebé en camino!
Serán necesarias al menos tres ecografías durante todo el embarazo, una por cada trimestre del embarazo. Pero es la primera ecografía la que más expectación genera, la que más ilusión hace, pero también la que más nervios produce en la mamá. ¿Estás preparada para tu primera ecografía? En Guiainfantil.com te contamos cuándo se hace, para qué sirve, qué verás en ella, etc. ¡Felicidades por tu bebé en camino!
Cuando una mujer se queda embarazada, cuando ve el resultado positivo del test, ya se imagina a su futuro hijo. Se genera una ilusión por la buena noticia y un deseo y expectación por realizar la primera ecografía y poder escuchar su latido y ver a su bebé.
La primera ecografía del embarazo te confirma que una vida se está gestando en tu interior. Se trata de un momento muy importante y emotivo porque es una especie de presentación de tu bebé. Por fin vas a conocer a tu bebé y, a través de la pantalla, ese primer contacto con tu hijo se hará inolvidable.
La primera ecografía es la más importante del primer trimestre y, en ocasiones, la única que se hace en esta etapa. Se suele realizar entre la semana 12-13. Se utilizan las dos vías, la vía abdominal y la vía vaginal para poder valorar mejor la anatomía. Mediante esta ecografía datamos la gestación, según lo que mida el feto se recalcula la fecha de la última regla y se calcula la fecha probable de parto. Como mediciones importantes tenemos:
– CRL (longitud craneocaudal): nos mide el feto de cabeza a región caudal (donde acaba la columna vertebral).
– DBP (diámetro biparietal): medición que se realiza en la cabeza, que nos será de ayuda para datar el embarazo si la ecografía se realizase a partir de la semana 14.
– TN (traslucencia nucal): también conocido como el pliegue nucal, uno de los marcadores más importantes del Síndrome de Down y de otras enfermedades genéticas.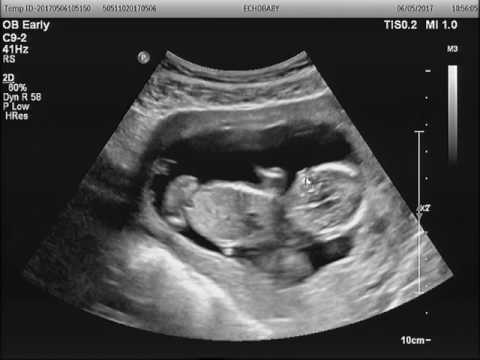
Cuando la mujer se da cuenta de que está embarazada normalmente es a partir de la semana 4 de gestación, cuando tiene la falta de la menstruación. A partir de ese momento la mujer está muy alerta para identificar signos y síntomas que puedan indicar que necesita una ecografía o una valoración por un ginecólogo/obstetra. Si presenta un sangrado, dolor abdominal, malestar… estos síntomas suelen ser una de las causas más típicas de consulta a urgencias.
La pregunta que se hacen la mayoría de las futuras mamás es ¿cuándo hay que hacerse la primera ecografía del embarazo? El deseo, por supuesto, es hacérsela cuanto antes, para comprobar que no hay complicaciones, que el bebé se desarrolla correctamente y que la gestación sigue adelante. Sin embargo, hay que tener en cuenta que, para comprobar todas esas cosas, debemos dar tiempo a que el embrión se vaya desarrollando.
¿Cuándo hacer la primera ecografía? Es una pregunta difícil ya que depende de muchas variables… Se sabe y ya se realiza en casi todo el mundo la ecografía del primer trimestre a las 12-13 semanas de embarazo, pero ¿es esa la primera ecografía que se realizan todas las embarazadas? En muchas ocasiones no, a veces se realiza antes y otras veces después.
– SEMANA 4:
Depende de en qué momento de la semana 4 de gestación estemos podemos desde no ver nada dentro del útero hasta ver un saco gestacional que se visualiza como una imagen negra circular dentro del útero. A saber, que el saco gestacional crece más o menos 1 mm al día.
– SEMANA 5:
En esta semana sí que debemos ver un saco gestacional y, además, en su interior se empezará a formar la vesícula vitelina que la podremos ver como un segundo anillo dentro del saco gestacional y suele medir entre 3-4 mm.
– SEMANA 6:
¡Ya se ve el embrión! A partir de esta semana ya sí que sí debemos de ver un embrión. Y se debería de ver también latido cardíaco. Pero muchas veces depende de lo que mida el embrión podremos o no ver el latido, está descrito que a partir de un embrión que mide 3.5mm se puede ver latido cardíaco, pero a veces no es hasta los 5-6mm que no apreciamos el latido. Lo que sí se sabe es que si el embrión ya mide 7mm y no vemos el latido probablemente se trate de una gestación no evolutiva.
Lo que sí se sabe es que si el embrión ya mide 7mm y no vemos el latido probablemente se trate de una gestación no evolutiva.
– SEMANA 7:
El embrión ya mide alrededor de 12mm, podemos diferenciar la cabeza, el cuerpo y los esbozos de las extremidades.
– SEMANA 8:
Si hacemos una ecografía estando de estas semanas el embrión medirá unos 20mm. Ya se pueden ver las extremidades formadas, el cordón umbilical y se aprecian los primeros movimientos.
– SEMANA 9:
En esta semana ya mide alrededor de 25mm y médicamente pasa a llamarse feto porque ya ha completado la organogénesis básica y desde ese momento los órganos irán madurando.
– SEMANA 10-12:
A partir de la semana 10 ya se pueden ver con forma humana, podemos diferenciar cabeza, tórax, abdomen, brazos y piernas y, en ocasiones, ver hasta los dedos, crecerán hasta medir unos 70mm.
[Leer más: El embarazo semana a semana]
La ecografía es una de las herramientas más importante para el seguimiento y control del embarazo. Pero centrándonos en el tema de hoy, ¿para qué nos sirve la ecografía en el primer trimestre? Para cinco cosas fundamentales:
1. Diagnosticar un embarazo.
2. Valorar viabilidad del embarazo:
Si a partir de las 6 semanas no viésemos un embrión o viésemos un embrión de unos 7mm sin latido, se trataría muy probablemente de un embarazo no viable.
3. Valorar la correcta implantación del embarazo:
Esto todavía no lo había mencionado, pero un 1.1 por ciento de los embarazos se localizan fuera del útero, se llaman embarazos ectópicos o extrauterinos. La gran mayoría se suelen encontrar en las trompas de Falopio, ovario o abdomen. Es importante el diagnostico de las gestaciones ectópicas porque pueden comportar un riesgo vital para la mujer, en muchos casos, su primer síntoma puede ser dolor abdominal o pequeño sangrado genital.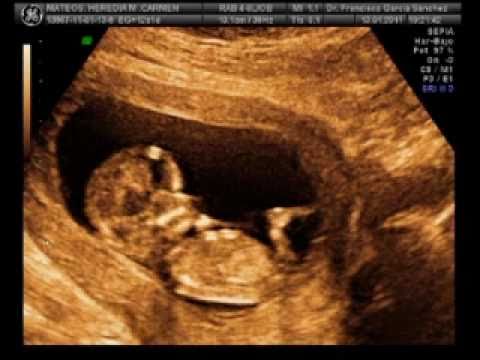
4. Número de embriones:
Si se trata de un embarazo múltiple, gemelar, trigemelar… Nos ayuda a contar el número de embriones y a saber si comparten bolsa, placenta o si cada uno tiene la suya propia.
5. Diagnostico de malformaciones:
En el primer trimestre se pueden diagnosticar algunas malformaciones fetales con un significado muy importante en su desarrollo futuro.
La primera ecografía es uno de los momentos más esperados de la mujer embarazada. Esta ecografía puede que se haga en cualquier momento del primer trimestre, dependiendo de cada caso, sea por algún síntoma (dolor abdominal, sangrado genital, malestar general…) que hace que la mujer consulte a urgencias o una ecografía programada. Durante el primer trimestre es cuando se empieza a formar el embrión, se pueden ver en función de la semana imágenes muy diferentes, por eso es muy importante la cautela, el conocimiento y ante cualquier duda realizar un nuevo control dejando un tiempo para valorar si el embarazo evoluciona favorablemente o no, no tomar decisiones precipitadas y explicar a la mujer la situación facilitando su compresión y aceptación.
Esperamos que te haya sido de utilidad este artículo. ¡Déjanos tus comentarios! ¡Te leemos!
Puedes leer más artículos similares a Emocionante primera ecografía del embarazo – Cuándo se hace y qué se ve, en la categoría de Etapas del embarazo en Guiainfantil.com.
R.F. Gainutdinova
Department of Ophthalmology of Kazan State Medical University
Kazan
Purpose: Study of efficacy of three-dimensional echography in combination with colored and energy Doppler mapping of blood flow in diagnostics of orbit pathology.
Materials and methods: Three-dimensional echography was performed in 25 patients (19 males and 6 females) with unilateral exophthalmos which were suspected to have space-occupying lesion of orbit. As a control 5 volunteers three –dimensional orbit echography. In all participants Doppler indices of ophthalmic artery (OA) ( V max OA, V min OA, V med OA, RI OA, PI OA, S/D OA) were detected on the main segment before forming arc by it above optic nerve.
Results: Malignant tumors are characterized by decrease of speed indices in veins and arteries of orbit because of their compression by tumor. PI OA is decreasing less then 1.12 and RI OA – less then 0.6. In pseudo tumors data of three-dimensional echography depends on prescription of diseases, localization and extent of process.
Conclusion: Three-dimensional echography in combination with colored and energy Doppler mapping of blood flow is a highly informative method of diagnostics of orbit pathology accompanied by exophthalmos.
Introduction
Improving the diagnosis of orbital pathology is one of the most difficult tasks in ophthalmology. The complexity of diagnosis is associated with a significant polymorphism of the pathology of the orbit, while the main and often the only clinical manifestation of it is exophthalmos. Among the radiation diagnostic methods used in identifying the cause of the pathology of the orbit, ultrasound imaging is one of the most informative. In recent years, thanks to the work of a number of researchers [4,6,7,8,9,10] in ophthalmology, such features of modern ultrasound technologies as three-dimensional echography with simultaneous Doppler blood flow mapping have been introduced. The diagnostic value of this method lies in the fact that it is possible to analyze the morphological structure of tissues with a simultaneous assessment of blood flow.
In recent years, thanks to the work of a number of researchers [4,6,7,8,9,10] in ophthalmology, such features of modern ultrasound technologies as three-dimensional echography with simultaneous Doppler blood flow mapping have been introduced. The diagnostic value of this method lies in the fact that it is possible to analyze the morphological structure of tissues with a simultaneous assessment of blood flow.
The purpose of this work was to study the effectiveness of the use of three-dimensional echography in combination with color (CDI) and energy (EC) Doppler blood flow mapping in the diagnosis of orbital pathology.
Material and methods
Three-dimensional echography was performed in 25 patients with unilateral exophthalmos (19 men and 6 women), who, when performing two-dimensional echography on the A / B scan sustems “Hymphrey inc.” there was a suspicion of a volumetric formation of the orbit. Three-dimensional echography was carried out on the device Voluson 730 “General Electric medical Systems”. In all cases of unilateral lesions, an ultrasound scan of the contralateral “healthy” orbit was taken as the norm. For control, three-dimensional echography of both orbits was performed on 5 healthy volunteers (three men and 2 women). Ultrasound parameters fully complied with the FDA recommendations of 09/30.97, as well as the provisions of the American Institute of Ultrasound in Medicine. Three-dimensional scanning was performed by a contact transpalpebral method through closed eyelids, using a special gel for ultrasound examination with a linear or convex probe with a frequency of 7.5–12 MHz.
In all cases of unilateral lesions, an ultrasound scan of the contralateral “healthy” orbit was taken as the norm. For control, three-dimensional echography of both orbits was performed on 5 healthy volunteers (three men and 2 women). Ultrasound parameters fully complied with the FDA recommendations of 09/30.97, as well as the provisions of the American Institute of Ultrasound in Medicine. Three-dimensional scanning was performed by a contact transpalpebral method through closed eyelids, using a special gel for ultrasound examination with a linear or convex probe with a frequency of 7.5–12 MHz.
Initially, at the first stage of the examination, the location of the eyeball and orbital structures, such as the optic nerve, extraocular muscles, and orbital fat, were visualized in a two-dimensional mode. Then 3D echography of the orbit was performed. Due to the unique function of the Voluson 730 ultrasonic device to simultaneously display the region of interest in three mutually perpendicular planes, we had the possibility of volumetric reconstruction and layer-by-layer study of the contents of the orbit. Further, using volumetric Doppler modes, a complete spectral analysis of the blood vessels of the study area was performed. Dopplerographic parameters of the ophthalmic artery (PA) were determined in all subjects on the segment of the main trunk of the artery until it formed an arc over the optic nerve. Qualitative and quantitative characteristics of hemodynamics in GA were assessed: maximum systolic blood flow velocity (Vmax HA) in cm/s, end-diastolic blood flow velocity (Vmin HA) in cm/s, mean blood flow velocity over the cardiac cycle (Vmed HA) in cm/s, resistivity index (RI GA), pulsation index (PI GA), systolic-diastolic ratio (S/D GA). In view of the great variability in the structure of the branches of the ophthalmic vein, the blood flow was determined and Dopplerometry of the superior ophthalmic vein was performed, which was visualized in the upper-medial region slightly above, anteriorly and medially from the place of registration of the GA. In the superior ophthalmic vein, the blood flow velocity was determined in cm/s.
Further, using volumetric Doppler modes, a complete spectral analysis of the blood vessels of the study area was performed. Dopplerographic parameters of the ophthalmic artery (PA) were determined in all subjects on the segment of the main trunk of the artery until it formed an arc over the optic nerve. Qualitative and quantitative characteristics of hemodynamics in GA were assessed: maximum systolic blood flow velocity (Vmax HA) in cm/s, end-diastolic blood flow velocity (Vmin HA) in cm/s, mean blood flow velocity over the cardiac cycle (Vmed HA) in cm/s, resistivity index (RI GA), pulsation index (PI GA), systolic-diastolic ratio (S/D GA). In view of the great variability in the structure of the branches of the ophthalmic vein, the blood flow was determined and Dopplerometry of the superior ophthalmic vein was performed, which was visualized in the upper-medial region slightly above, anteriorly and medially from the place of registration of the GA. In the superior ophthalmic vein, the blood flow velocity was determined in cm/s. Then the presented three-dimensional image of the orbit was examined from any side of interest using the rotation function. When a volumetric formation was detected, using the “virtual scalpel” function, it was cut off along various planes, thereby specifying the localization of the pathological process, its internal structure, the state of the membranes, as well as the relationship with other structures of the orbit.
Then the presented three-dimensional image of the orbit was examined from any side of interest using the rotation function. When a volumetric formation was detected, using the “virtual scalpel” function, it was cut off along various planes, thereby specifying the localization of the pathological process, its internal structure, the state of the membranes, as well as the relationship with other structures of the orbit.
Results and Discussion
In each case, the study of a patient with exophthalmos got acquainted with the history of the disease, the results of an ophthalmological examination, laboratory data, the results of computed and magnetic resonance imaging (CT and MRI).
As a result of the study of three-dimensional echograms in 13 patients, in the absence of “plus-tissue”, an increase in the thickness of one or more muscles of the orbit, deformation of their fibrous membrane, an increase in the length of the retrobulbar space, and changes in its echogenicity were revealed. When mapping the blood flow of this group of patients, a large number of dilated vessels of the orbit were revealed (Fig. 1). Dopplerometry determined an increase in blood flow velocity (Vmax GA) by 1.7–2.2 times (p<0.05) compared with healthy people, an increase in resistance indices (PI, RI, S/D) of GA by 1.2 times (p <0.05), which is typical for orbital pseudotumor with its protracted course [1,5]. A decrease in blood flow velocity in the ophthalmic veins by 1.3–2.5 times (p<0.05) was also revealed. The vessels were compressed by dilated muscles and edematous tissue of the orbit. Analyzing the data of anamnesis, clinical manifestations and ultrasound examinations in this subgroup, a diagnosis of pseudotumor, primary idiopathic myositis of the orbit, was made. It should be noted that GA Doppler parameters depended on the stage of the disease.
When mapping the blood flow of this group of patients, a large number of dilated vessels of the orbit were revealed (Fig. 1). Dopplerometry determined an increase in blood flow velocity (Vmax GA) by 1.7–2.2 times (p<0.05) compared with healthy people, an increase in resistance indices (PI, RI, S/D) of GA by 1.2 times (p <0.05), which is typical for orbital pseudotumor with its protracted course [1,5]. A decrease in blood flow velocity in the ophthalmic veins by 1.3–2.5 times (p<0.05) was also revealed. The vessels were compressed by dilated muscles and edematous tissue of the orbit. Analyzing the data of anamnesis, clinical manifestations and ultrasound examinations in this subgroup, a diagnosis of pseudotumor, primary idiopathic myositis of the orbit, was made. It should be noted that GA Doppler parameters depended on the stage of the disease.
Further examination revealed thyroid pathology in the form of diffuse toxic goiter (3), autoimmune thyroiditis (4 patients) in 7 patients from this subgroup, and endocrine myopathy was diagnosed. It is well known that the main sign of differential diagnosis of endocrine ophthalmopathy is the bilateral nature of the disease. But often, as described in the literature [2,3], the interval between lesions of paired orbits can be several years (as was observed in the patients we examined).
It is well known that the main sign of differential diagnosis of endocrine ophthalmopathy is the bilateral nature of the disease. But often, as described in the literature [2,3], the interval between lesions of paired orbits can be several years (as was observed in the patients we examined).
15 patients with space-occupying orbital masses identified by 2D scanning underwent 3D echography. According to the recommendations [5], proposed for use in echographic detection of a tumor, the topography of the mass formation, shape and contours, dimensions, its prevalence, internal echostructure of the tumor were determined. These echographic parameters, which were also determined by three-dimensional echography, made it possible to suggest the nature of tumor-like formations. Depending on the nature of echo signals, tumors were divided into solid, cystic, vascular, and infiltrative types. Most of the benign tumors we identified, according to this division, manifested themselves on the scans as a solid type – these are lipoma (1), meningioma (2), neurinoma (1), fibroma (2). On scans, they were detected as echopositive formations of a round or oval shape, with clear contours, with an ordered internal structure, without a pronounced capsule. Cystic types of formations – dermoid cyst (3) – had an echo-negative internal structure, a clearly differentiated capsule, and regular shapes. Vascular tumors – hemangiomas (2) – on ultrasound scans were determined as an irregular shape, with clear uneven contours of the formation with many partitions in the structure, increased in size under stress. At the next stage of the ultrasound study, orbital blood flow was mapped and the presence or absence of orbital vessel deformity was determined, changes in hemodynamics were assessed, and the presence of blood flow inside the tumor was determined. CDI and EC of the orbital vessels in patients with a malignant tumor (2) revealed the presence of their own vascular network, deformation of the vascular pattern and (or) a decrease in the speed indicators in the arteries and veins of the orbit due to their compression by the tumor.
On scans, they were detected as echopositive formations of a round or oval shape, with clear contours, with an ordered internal structure, without a pronounced capsule. Cystic types of formations – dermoid cyst (3) – had an echo-negative internal structure, a clearly differentiated capsule, and regular shapes. Vascular tumors – hemangiomas (2) – on ultrasound scans were determined as an irregular shape, with clear uneven contours of the formation with many partitions in the structure, increased in size under stress. At the next stage of the ultrasound study, orbital blood flow was mapped and the presence or absence of orbital vessel deformity was determined, changes in hemodynamics were assessed, and the presence of blood flow inside the tumor was determined. CDI and EC of the orbital vessels in patients with a malignant tumor (2) revealed the presence of their own vascular network, deformation of the vascular pattern and (or) a decrease in the speed indicators in the arteries and veins of the orbit due to their compression by the tumor. Thanks to the special programs that the Voluson 730 three-dimensional echography apparatus is equipped with, tissue was cut along the tumor, its cross section was obtained, by which it was possible to assess the internal structure, the presence of vascularization of the neoplasm, the degree of germination in the surrounding tissues.
Thanks to the special programs that the Voluson 730 three-dimensional echography apparatus is equipped with, tissue was cut along the tumor, its cross section was obtained, by which it was possible to assess the internal structure, the presence of vascularization of the neoplasm, the degree of germination in the surrounding tissues.
Figure 2 shows a three-dimensional echogram of the patient Sh. with an optic nerve tumor in combination with energy mapping of blood flow. The scanning plane runs along the axis of the optic nerve and neoplasm and reveals the location of the tumor eccentric relative to the optic nerve, the absence of vascularization inside the tumor, which is typical for a benign tumor of the optic nerve – meningioma.
In two patients, three-dimensional echography in the CDI and EC modes inside the tumor revealed a blood flow with a low-amplitude nature of the collateral type with a decrease in pulsation and resistivity indices, as well as deformation of the vascular pattern and a decrease in velocity indicators in the arteries and veins of the orbit due to their compression by the tumor. After analyzing the results of three-dimensional echography, CT and MRI, as well as anamnesis and clinical manifestations, a malignant tumor of the orbit was diagnosed (Fig. 3).
After analyzing the results of three-dimensional echography, CT and MRI, as well as anamnesis and clinical manifestations, a malignant tumor of the orbit was diagnosed (Fig. 3).
In two examined patients with exophthalmos, two-dimensional echography revealed hypoechoic formations in the form of strands with clear contours that did not deform the eyeball (Fig. 4).
Blood flow mapping made it possible to establish that they were represented by dilated veins with a low blood flow velocity (Fig. 5). Three-dimensional echography visualized the presence of a thrombus in the lumen of the main vessel trunk. Thus, summing up the anamnestic, ophthalmological, laboratory and ultrasound data, a diagnosis of orbital thrombophlebitis was made. Repeated echography on the background of treatment stated a positive trend.
Conclusions
1. The high diagnostic informativeness of three-dimensional echography in combination with color doppler and EC of orbital pathology has been established, which determines the possibility of using it in the differential diagnosis of orbital diseases accompanied by exophthalmos.
2. An orbital tumor is characterized by the detection of a volumetric formation, the echographic characteristics of which depend on the nature of the tumor, the presence of deformation of the vascular pattern and (or) a decrease in speed indicators in the arteries and veins of the orbit due to their compression by the tumor (this is especially pronounced in patients with a malignant tumor). A malignant tumor is also characterized by visualization of blood flow inside the tumor with a low-amplitude nature of the collateral type with a decrease in indices – PI HA below 1.12 and RI HA below 0.6.
3. With pseudotumor, the data of three-dimensional echography depend on the duration of the disease, localization and prevalence of the process. The stage of infiltration is characterized by an increase in the length of the retrobulbar zone and (or) thickening of one or a group of orbital muscles, increased blood flow with a significant increase in GA Vmax and a decrease in hemodynamic resistance. The stage of transition to fibrosis is characterized by thickening and deformation of the orbital structures, a decrease in GA Vmin, an increase in GA S/D, GA PI, and GA RI.
The stage of transition to fibrosis is characterized by thickening and deformation of the orbital structures, a decrease in GA Vmin, an increase in GA S/D, GA PI, and GA RI.
4. With thrombophlebitis of the orbit, hypoechoic formations are visualized in the form of strands with even clear contours that do not deform the eyeball. Three-dimensional echography with color doppler and EC revealed that they were represented by dilated veins with thickened walls and low blood flow velocity.
Literature
1. Aznabaev M. T. The role of the hemodynamic factor in the diagnosis of tumors and tumor-like diseases of the orbit / M. T. Aznabaev, A. F. Gabdrakhmanova / Modern methods of radiation diagnostics: materials of scientific–practical. Conf. dedicated to the 60th anniversary of the Russian Academy of Medical Sciences. – M., 2004. – S. 168-170.
2. Brovkina A. F. Diseases of the orbit / A. F. Brovkina. – M.: Medicine, 1993. – 240 p.
3. Brovkina A. F. Endocrine ophthalmopathy / A. F. Brovkina. – M.: GEOTAR-MED, 2004. – 174 p.
F. Endocrine ophthalmopathy / A. F. Brovkina. – M.: GEOTAR-MED, 2004. – 174 p.
4. Kruglova E. V. Three-dimensional ultrasound reconstruction of the vessels of the eye and orbit: author. dis. E cand. honey. Sciences / E. V. Kruglova. – M., 2003. – 24 p.
5. Complex ultrasonic diagnostics of volumetric formations of the orbit: guidelines / M. T. Aznabaev [et al.] – Ufa, 2001. – 10 p.
6. Nasnikova I. Yu. Spatial ultrasound diagnosis of diseases of the eye and orbit. Clinical guidance / I. Yu. Nasnikova, S. I. Kharlap, E. V. Kruglova. – M., 2004. – 174 p.
7. Nasnikova I. Yu. Ultrasound volumetric spatial imaging and the possibility of its use in ophthalmology / I. Yu. Nasnikova, S. I. Kharlap // Medical visualization. – 2003. – No. 3. – P. 49–58.
8. Nasnikova I. Yu. New diagnostic possibilities in ultrasound examination of the eye and orbit / I. Yu. Nasnikova, S. I. Kharlap, E. V. Kruglova // Echography. – 2002. – No. 3. – S. 236-241.
9. Fundamentals of spatial ultrasound visualization of the eye and orbit / S. E. Avetisov [et al.] / Modern methods of radiation diagnostics: materials of scientific–practical. Conf. dedicated to the 60th anniversary of the Russian Academy of Medical Sciences. – M., 2004. – S. 14–25.
E. Avetisov [et al.] / Modern methods of radiation diagnostics: materials of scientific–practical. Conf. dedicated to the 60th anniversary of the Russian Academy of Medical Sciences. – M., 2004. – S. 14–25.
10. Three-dimensional ultrasonic reconstruction of the angioarchitectonics of the anatomical structures of the eye and orbit / V. S. Akopyan [et al.] // Kremlin medicine. Clinical Bulletin. – 2002. – No. 2. – P. 54–57.
In order to study the normal echographic anatomy of the scrotum, a polypositional ultrasound examination was performed in 219 children without pathology of the inguinal-scrotal region aged from 1 month to 18 years. The technique of ultrasound examination of the scrotum organs is given, the main anatomical structures are indicated. The age standards for the size of the scrotum organs in children are presented.
Fig. 1. Echogram of the scrotum wall of an 11-year-old child. Longitudinal scan. 1 – protein shell, 2 – fleshy shell, 3 – skin
Longitudinal scan. 1 – protein shell, 2 – fleshy shell, 3 – skin
Fig. 2. Echogram of the scrotum of a 1 year old child. Cross scan. The arrow indicates the median septum of the scrotum
Fig. 3. Echogram of the testis of a child of 11 years old. Longitudinal scan. 1 – testicle, 2 – head of the epididymis
Fig. 4. Echogram of the scrotum of a 3-year-old child with dropsy of the testicular membranes. 1 – testicle, 2 – albuginea, 3 – dropsy fluid
Fig. 5. Echogram of the unaltered testicle of an 8-year-old child. 1 – testicle in cross section, 2 – liquid contents of the serous cavity
Fig. 6. Echogram of the unaltered testicle of a 10-year-old child. Color Doppler mapping mode. 1 – testicle in cross section, 2 – testicular vessels
Fig. 7. Testicle and its appendage. 1 – spermatic cord, 2 – head of the epididymis, 3 – appendage of the epididymis, 4 – ligament of the epididymis, 5 – upper end of the testicle, 6 – appendix of the testicle, 7 – albuginea, 8 – front edge of the testicle, 9 – lower end egg
Fig. 8. Schematic representation of the normal anatomy of the scrotum. 1 – convoluted tubules, 2 – direct tubules, 3 – galley network, 4 – maxillary body, 5 – efferent tubules, 6 – head of the appendage, 7 – body of the appendage, 8 – tail of the appendage, 9– deviating
8. Schematic representation of the normal anatomy of the scrotum. 1 – convoluted tubules, 2 – direct tubules, 3 – galley network, 4 – maxillary body, 5 – efferent tubules, 6 – head of the appendage, 7 – body of the appendage, 8 – tail of the appendage, 9– deviating
Fig. Fig. 9. Echogram of the testis of a 16-year-old patient: a – transverse echogram, b – longitudinal echogram. Mediastinum indicated by arrows
Table 1. Main testicular parameters in children of different age groups (n = 219)
Fig. 10. Echogram of the testicles of an 11-year-old child is normal. 1 – testicle, 2 – head of the epididymis
Fig. Fig. 11. Echogram of the unaltered testicle and its epididymis in a 16-year-old child: a – transverse scan, b – oblique scan. The arrows indicate the body of the epididymis
Table 2. Main parameters of the head of the epididymis (mm) in children of different age groups (n = 219)
Fig. 12. Echogram of the unchanged testicle and tail of the epididymis.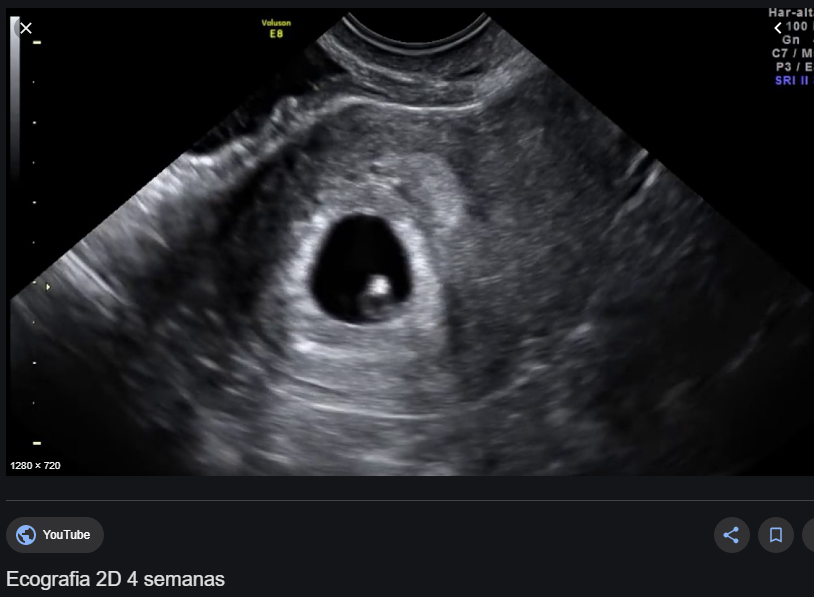 Longitudinal scan. 1 – testicle, 2 – vas deferens, 3 – tail of the epididymis
Longitudinal scan. 1 – testicle, 2 – vas deferens, 3 – tail of the epididymis
Fig. 13. Scrotal part of the spermatic cord of an 11-year-old patient. Longitudinal scan. The arrows indicate the vas deferens. 1 – testicle, 2 – spermatic cord
Fig. 14. Deferent duct: a – echogram of the scrotal part of the spermatic cord. The arrows indicate the spermatic cord, (b) the echogram of the vas deferens and the tail of the epididymis. 1 – testicle, 2 – tail of the appendage. The arrows indicate the vas deferens
Fig. 15. Variants of visualization of the unchanged hydatid. Hydatida indicated by arrows
Fig. 16. Echogram of the testis of a 10-year-old child. CDI mode. The testicular artery is indicated by arrows. Along the spermatic cord, the testicular artery is stained in red (the flow is directed to the sensor), in the posterolateral part of the testicle – in blue (the flow is directed
Table 3. Normal testicular blood flow parameters in children (M ± m)
Fig. 17. Echogram of the testis of a 10-year-old child. CDI mode. Arrows indicate a large branch of the capsular artery. 20.tif
17. Echogram of the testis of a 10-year-old child. CDI mode. Arrows indicate a large branch of the capsular artery. 20.tif
Fig. 18. Echogram of the testis of a 10-year-old child. CDI mode. Centropetal arteries stained blue
Fig. 19. Echogram of the testis of an 11-year-old child. Triplex mapping. Centropetal arteries stained blue
Fig. 20. Echogram of the testis of a 10-year-old child. CDI mode. Centropetal arteries are stained in red, recurrent arteries in blue
Fig. 21. Color Doppler mapping of testicular vessels in a 14-year-old child
Fig. 22. Echogram of the testis of a 13-year-old child. CDI mode. Transmediastenal artery
Fig. 23. Echogram of the testis of a 13-year-old child. CDI mode. Parallel to the transmediastinal artery (red) is a vein (blue)
Fig. 24. Echogram of the testis of a 13-year-old child. CDI mode. Transmediastinal vein carrying blood from the peripheral parts of the testis to the testicular mediastinum
Fig. 25. Echogram of the testis of a 13-year-old child. TsDK and impulse-wave dopplerography. Intratesticular vein
25. Echogram of the testis of a 13-year-old child. TsDK and impulse-wave dopplerography. Intratesticular vein
Fig. 26. Echogram of the pampiniform plexus in a 4-year-old child
Fig. 27. Echogram of the unaltered pampiniform plexus in a 15-year-old child
Introduction
Diseases associated with impaired obliteration of the vaginal process of the peritoneum, cryptorchidism, varicocele most often require planned surgical treatment in pediatric practice. Congenital inguinal hernias are diagnosed in 1-5% of children, which is 92-95% of all types of hernias in childhood, hydrocele – in 4.6% [1, 2]. Operations on the inguinal canal and testis in children account for more than 67% of all planned surgical interventions [2]. It is known that approximately 58% of diseases of childhood may pose a threat of infertility for men in the future. The reproductive health of boys depends both on the timely diagnosis of diseases of the scrotum, the jewelry technique for performing operations on the inguinal canal, testicles, penis, and on the accurate conduct of the postoperative, rehabilitation, dispensary periods.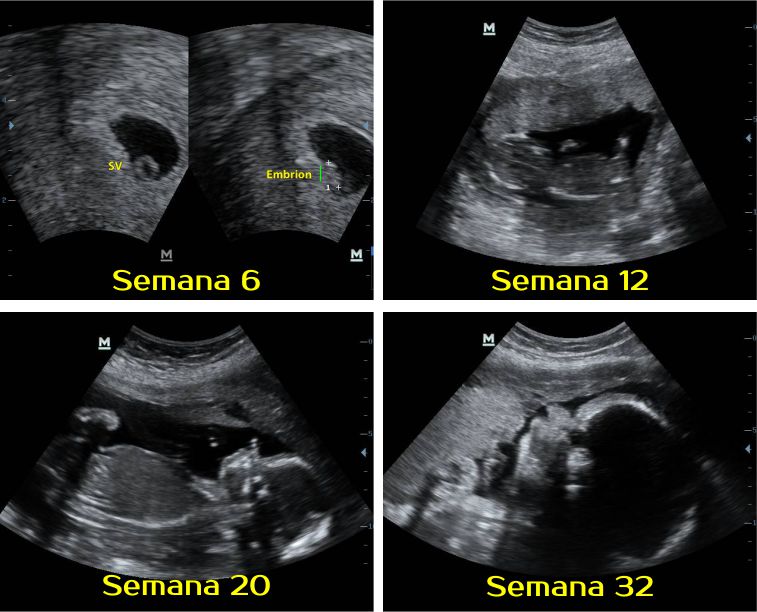 With the advent of modern diagnostic techniques, preference is given to non-invasive research methods.
With the advent of modern diagnostic techniques, preference is given to non-invasive research methods.
Numerous monographs and scientific articles devoted to the diagnosis of diseases of the scrotum organs describe in detail the methods of ultrasound examination of the scrotum organs [3–18]. Despite the apparent simplicity and accessibility of ultrasound examination of the scrotum, fundamental questions remain regarding both the ultrasound technique itself and the diagnosis of certain diseases of the reproductive system. In practice, most ultrasound diagnostic specialists refuse to examine the scrotum for various reasons. At the same time, cases of gross diagnostic errors have become more frequent, when radiologists, not having sufficient experience in examining the organs of the reproductive system, issue incorrect conclusions. The aim of our work was to develop a methodology for ultrasound examination of the scrotum organs and to study the age standards for the size of the scrotum organs in children of different age groups.
Research materials and methods
An analysis of a comprehensive ultrasound examination of 219 children without pathology of the inguinal-scrotal region aged from 1 month to 18 years, who were examined and treated in the surgical departments of the Children’s Clinical Hospital of Tver in the period from 2000 to 2008 d. The study was conducted for all children on HD-11XE and iU-22 ultrasonic devices (Philips, the Netherlands). Broadband linear transducers with a scanning frequency of 5–12 MHz in gray scale mode were used, as well as color and power Doppler mapping, and pulsed wave Dopplerography.
No prior preparation for the study was required. The study was performed with the patient lying on his back with slightly bent legs apart. Scanning of the scrotum was performed in several planes: transverse, oblique, longitudinal, oblique. Initially, optimal B-mode ultrasound images of the testis, epididymis, scrotal structures of the spermatic cord, and scrotal wall were obtained. In this mode, the dimensions, volume (V = length × width × thickness × 0.523), shape, echogenicity, and homogeneity of the gonadal structure were determined.
In this mode, the dimensions, volume (V = length × width × thickness × 0.523), shape, echogenicity, and homogeneity of the gonadal structure were determined.
Color Doppler ultrasonography (CDO) made it possible to assess the degree of testicular vascularization by the number of vascular signals. With pulsed wave Doppler sonography, absolute indicators (peak systolic and end-diastolic blood flow velocity) and relative quantitative indicators (resistance index) were studied. The parameters of blood flow in the testicular (along the scrotal part of the spermatic cord), capsular, centropetal and recurrent arteries were studied. The results obtained were processed by standard statistical methods. Quantitative data depending on the nature of the distribution are presented as M ± m, median, 5–95th percentile. Differences were considered significant at p ≤ 0.05.
Research results and discussion
Knowledge of anatomy is an important condition for ultrasound examination of the scrotum. The scrotum is a musculocutaneous organ consisting of two symmetrical halves, each containing a testis, an epididymis, and the scrotal part of the spermatic cord [19, 20]. The wall of the scrotum is formed by the seven layers of the testis. The skin of the scrotum is an extension of the skin of the penis. It is thin, has no fat, wrinkled, and is defined by ultrasound as a thin hyperechoic structure. Under the skin is a fleshy membrane (tunica dartos), which consists of smooth muscle fibers, the contraction of which causes the formation of folds in the skin of the scrotum. The fleshy membrane is visualized as a less echogenic, but more pronounced structure of the scrotal wall (Fig. 1). The next layers are the outer seminal fascia; fascia of the muscle that lifts the testicle; the internal seminal fascia and the vaginal membrane of the testis, consisting of parietal and visceral sheets. The wall thickness of the scrotum normally ranges from 2 to 8 mm, depending on the severity of the fleshy membrane.
The scrotum is a musculocutaneous organ consisting of two symmetrical halves, each containing a testis, an epididymis, and the scrotal part of the spermatic cord [19, 20]. The wall of the scrotum is formed by the seven layers of the testis. The skin of the scrotum is an extension of the skin of the penis. It is thin, has no fat, wrinkled, and is defined by ultrasound as a thin hyperechoic structure. Under the skin is a fleshy membrane (tunica dartos), which consists of smooth muscle fibers, the contraction of which causes the formation of folds in the skin of the scrotum. The fleshy membrane is visualized as a less echogenic, but more pronounced structure of the scrotal wall (Fig. 1). The next layers are the outer seminal fascia; fascia of the muscle that lifts the testicle; the internal seminal fascia and the vaginal membrane of the testis, consisting of parietal and visceral sheets. The wall thickness of the scrotum normally ranges from 2 to 8 mm, depending on the severity of the fleshy membrane. The median septum divides the scrotum into two halves (Fig. 2).
The median septum divides the scrotum into two halves (Fig. 2).
The testicle is covered with a thin connective tissue membrane called the tunica albuginea. In the testicle, the inner and outer surfaces, the anterior and posterior edges, the upper and lower poles are distinguished. Along the posterior edge of the testicle is its appendage (epididymis). The testicle and appendage are surrounded by a vaginal membrane (tunica vaginalis testis), which forms a serous cavity around them. On longitudinal scanning, the testicle has an oval shape with a clear, even contour (Fig. 3). The echogenicity of the testis is normally medium, the structure is fine-grained. The use of high-frequency transducers, as well as the presence of fluid in the serous cavity of the scrotum, make it possible to visualize the albuginea as a thin hyperechoic structure around the testicle (Fig. 4). In children, especially in the younger age group, the serous cavity contains a small amount of fluid, which is defined as a thin anechoic zone 1–3 mm wide (Fig. 5).
5).
The testicle is an intraperitoneal organ and is covered by a visceral plate that passes along the posterior edge of the testicle into the parietal plate. The parietal plate is firmly fused with the albuginea of the testicle to its posterior edge, where there is an area not covered by the parietal plate, through which nerves and vessels enter the testicular parenchyma (Fig. 6). The testicle is suspended on the spermatic cord (the left one is lower than the right one) in such a way that it is tilted with its upper end forward, and with its lateral surface somewhat backward [19]. The spermatic cord is defined from behind and from above (Fig. 7) [19]. From the albuginea into the parenchyma of the testis, the testicular septa (septula testis) extend radially, which divide the gonad into lobules (lobuli testis). The septa in the posterior superior region unite and form the testicular mediastinum (Fig. 8) [21]. Normally, the number of lobules ranges from 100 to 250.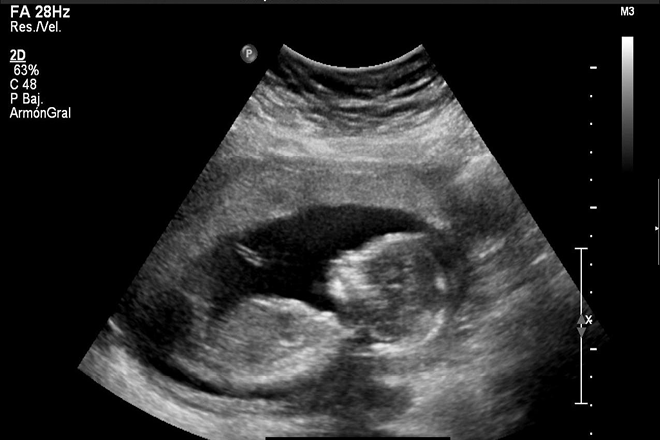 Each lobule contains 3–4 seminiferous tubules, each of which is 70–100 cm long. From this network, up to 18 efferent tubules of the testis are formed, which flow into the ducts of the head of the epididymis (vas efferent). In ultrasound examination, the testicular mediastinum is visualized along its posterior-superior surface, has a triangular shape and higher echogenicity than the surrounding parenchyma (Fig. 9). The dimensions of the testicles are measured in three mutually perpendicular planes. The main parameters of the testicles, obtained by us in the course of the study, are shown in Table 1.
Each lobule contains 3–4 seminiferous tubules, each of which is 70–100 cm long. From this network, up to 18 efferent tubules of the testis are formed, which flow into the ducts of the head of the epididymis (vas efferent). In ultrasound examination, the testicular mediastinum is visualized along its posterior-superior surface, has a triangular shape and higher echogenicity than the surrounding parenchyma (Fig. 9). The dimensions of the testicles are measured in three mutually perpendicular planes. The main parameters of the testicles, obtained by us in the course of the study, are shown in Table 1.
The epididymis (epididymis) is a connecting organ between the testicle and the vas deferens. It is located on the posterior edge of the testis. The appendage performs not only a transport function, but also contributes to the maturation of spermatozoa. Anatomically, there are three parts in the appendage: the head of the appendage (caput epididymidis), the body of the appendage (corpus epididymidis) and the tail of the appendage (cauda epididymidis). The head of the epididymis is located in the upper part of the testicle, protrudes above the upper pole, has a triangular shape. The body of the appendage occupies the middle part and is the thinnest structure, 2–4 mm thick. The tail of the epididymis connects the epididymis with the vas deferens [19]. Normally, ultrasound examination in all age groups clearly differentiates the head of the epididymis. Its structure and echogenicity are comparable to those of the testicular parenchyma (Fig. 10). Measurements of the head of the epididymis are carried out in the sagittal plane, while two sizes are sufficient – anterior-posterior and vertical (Table 2). Visualization of the body of the appendage is difficult due to its small size and location. However, in children of the older age group, it is possible to visualize the body of the appendage during transverse scanning, as well as when scanning at an angle, when the sensor is directed posteriorly and medially.
The head of the epididymis is located in the upper part of the testicle, protrudes above the upper pole, has a triangular shape. The body of the appendage occupies the middle part and is the thinnest structure, 2–4 mm thick. The tail of the epididymis connects the epididymis with the vas deferens [19]. Normally, ultrasound examination in all age groups clearly differentiates the head of the epididymis. Its structure and echogenicity are comparable to those of the testicular parenchyma (Fig. 10). Measurements of the head of the epididymis are carried out in the sagittal plane, while two sizes are sufficient – anterior-posterior and vertical (Table 2). Visualization of the body of the appendage is difficult due to its small size and location. However, in children of the older age group, it is possible to visualize the body of the appendage during transverse scanning, as well as when scanning at an angle, when the sensor is directed posteriorly and medially.
In this case, it is possible to visualize it throughout (Fig. 11). The tail of the epididymis is determined by the lower pole of the testicle, it is more elongated, the structure is somewhat heterogeneous due to the tortuosity of the vas deferens, and its echogenicity is lower than that of the head of the epididymis and testicular parenchyma (Fig. 12). Measurements of the linear dimensions of the body and tail of the epididymis are not fundamental, although in pathology they increase in size, acquire a different echogenicity depending on the nature of the pathology, which should be reflected in the study protocol. The vas deferens (ductus deferens) is a long tube (up to 50 cm) with a diameter of up to 3 mm (Fig. 13-14). Hydatids (appendix testis), being derivatives of the primary genital ducts, often persist in males in the form of pendants on the testicle or epididymis. Hydatida is visualized more often as an oval-shaped formation, fixed to the testicle or epididymis, of a homogeneous structure, of medium echogenicity. Hydatids vary in size from 2 to 5–7 mm.
11). The tail of the epididymis is determined by the lower pole of the testicle, it is more elongated, the structure is somewhat heterogeneous due to the tortuosity of the vas deferens, and its echogenicity is lower than that of the head of the epididymis and testicular parenchyma (Fig. 12). Measurements of the linear dimensions of the body and tail of the epididymis are not fundamental, although in pathology they increase in size, acquire a different echogenicity depending on the nature of the pathology, which should be reflected in the study protocol. The vas deferens (ductus deferens) is a long tube (up to 50 cm) with a diameter of up to 3 mm (Fig. 13-14). Hydatids (appendix testis), being derivatives of the primary genital ducts, often persist in males in the form of pendants on the testicle or epididymis. Hydatida is visualized more often as an oval-shaped formation, fixed to the testicle or epididymis, of a homogeneous structure, of medium echogenicity. Hydatids vary in size from 2 to 5–7 mm. More often, the hydatid attaches to the superior pole of the testicle or to the sulcus where the testicle and epididymis meet [19].]. Hydatids often consist of several lobules, more often they are two-lobed (Fig. 15).
More often, the hydatid attaches to the superior pole of the testicle or to the sulcus where the testicle and epididymis meet [19].]. Hydatids often consist of several lobules, more often they are two-lobed (Fig. 15).
Normal testicular blood flow parameters
Blood supply to the scrotum is provided by the internal pudendal artery of the levator testicular muscle (a. сremasterica). Venous outflow occurs through the external pudendal veins and the pampiniform plexus of the veins of the spermatic cord (plexus pampiniformis). The organs of the scrotum are supplied with blood by the following vessels: the testicular artery, the artery of the vas deferens, the cremaster artery, the anterior and posterior scrotal arteries, and the perineal artery. Other arteries are branches of the internal and external iliac arteries. The listed vessels anastomose widely among themselves, providing a good blood supply to the scrotum. The testicular artery penetrates the parenchyma, in the projection of the posterolateral sections of the upper pole of the testicle is divided into capsular arteries [19].
In the color Doppler mode, signals from the testicular artery and capsular branches can be easily recorded in all age groups (Fig. 16–17). The testicular artery is visualized at the upper pole of the testicle along its posterior outer surface (Fig. 16). The capsular artery is defined superficially around the testicle, along the albuginea. Centropetal arteries originate from the capsular arteries and run towards the testicular mediastinum (Fig. 18-19). Before reaching the mediastinum, the centropetal arteries continue into the recurrent arteries, carrying blood in the opposite direction (Fig. 20). The centropetal and recurrent arteries show different colors on color Doppler imaging. It is possible to register both types of vessels, as a rule, starting from the age of 10–11 years. In children of the younger age group, the capsular and centropetal arteries extending from them are well visualized (Fig. 21).
In more than 50% of cases, the testicular artery gives rise to a large branch, the transmediastinal artery, which runs from the mediastinum in the direction opposite to the direction of blood flow in the centropetal arteries (Fig. 22). The parameters of intratesticular circulation were studied according to the results of pulsed wave Doppler sonography. In color and power Doppler mapping, the distribution of vascular signals in the testicular parenchyma was uniform on both sides. The results obtained by us are reflected in Table 3. Venous outflow is carried out by veins of two types: veins extending from deep-seated parts of the testicle, and veins draining the superficial zones of the organ. Visualization of intratesticular veins is very difficult due to low speed indicators, but sometimes it is possible to determine them. Most often, intratesticular veins are visualized, going towards the testicular mediastinum, accompanying the transmediastinal arteries (Fig. 23-25).
22). The parameters of intratesticular circulation were studied according to the results of pulsed wave Doppler sonography. In color and power Doppler mapping, the distribution of vascular signals in the testicular parenchyma was uniform on both sides. The results obtained by us are reflected in Table 3. Venous outflow is carried out by veins of two types: veins extending from deep-seated parts of the testicle, and veins draining the superficial zones of the organ. Visualization of intratesticular veins is very difficult due to low speed indicators, but sometimes it is possible to determine them. Most often, intratesticular veins are visualized, going towards the testicular mediastinum, accompanying the transmediastinal arteries (Fig. 23-25).
Both types of testicular veins merge to form the pampiniform plexus outside the testicle, forming the testicular vein, which flows into the inferior vena cava on the right and into the left renal vein on the left. The vas deferens vein and the levator vein originate from the venous plexuses of the same name.